What to know about Covid-19 vaccines for young children
Sign up now: Get ST's newsletters delivered to your inbox
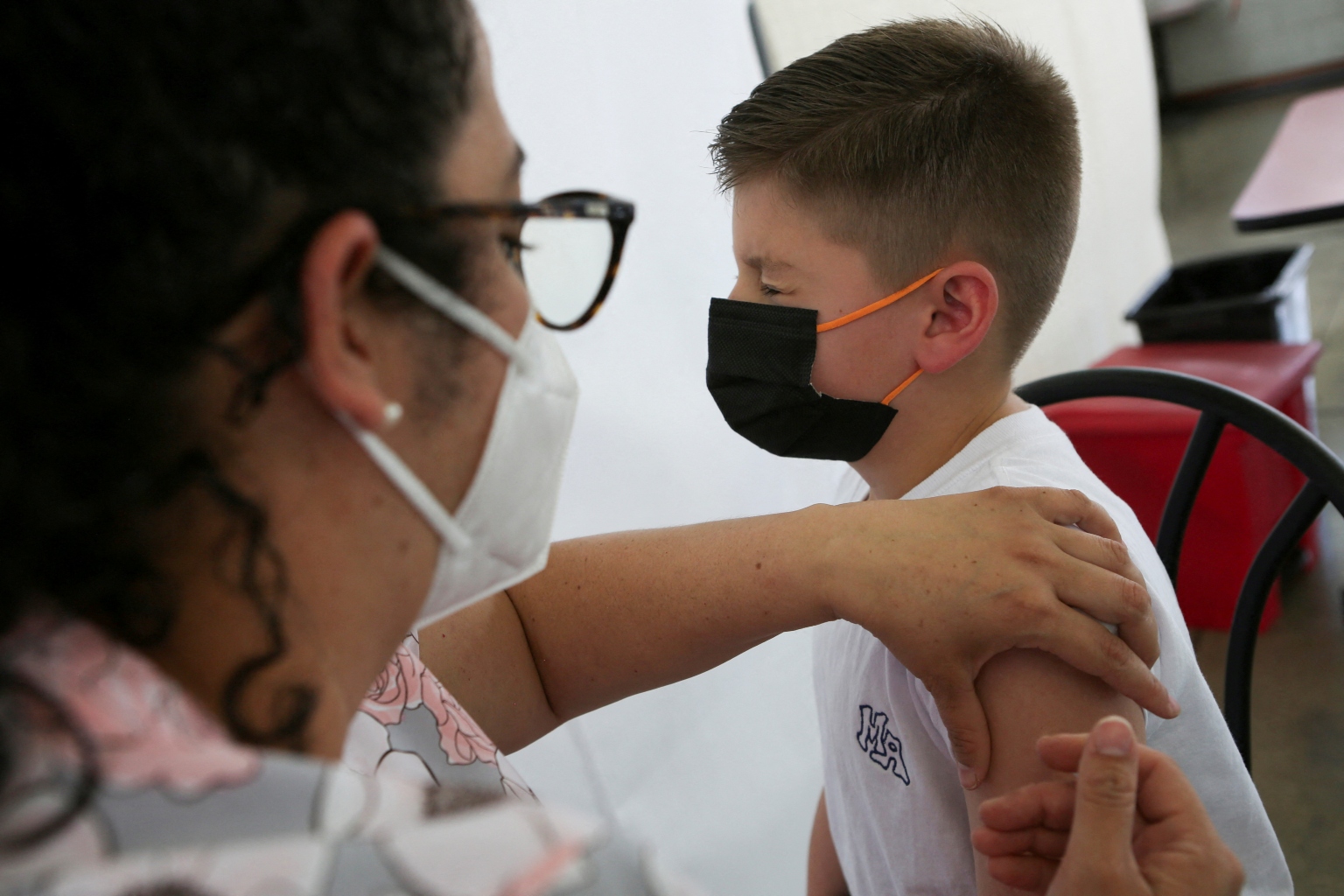
Neither the Moderna nor the Pfizer vaccine has raised any serious safety issues in the trials.
PHOTO: REUTERS
(REUTERS) - A panel of advisers to the US Food and Drug Administration (FDA) on Wednesday (June 15) unanimously voted to recommend Moderna's Covid-19 vaccine for children under six years old and Pfizer-BioNTech's for children under five.
The following are some important details to know about these two messenger RNA (mRNA) vaccines for the age groups.
How many doses are needed?
Moderna has asked the FDA to authorise a two-dose regimen of its vaccine in children aged six months to six years old. The 25mg doses are given 28 days apart. Adults receive 100mg per shot for their first two doses.
Pfizer and BioNTech are asking for a three-dose course of their vaccine for children aged six months to under five years old.
The first two doses are given 21 days apart and the third is administered at least two months after the second. Each dose is 3mg, below the 10mg given to children aged five to 11 and 30mg for people aged 12 and older.
Both companies sought lower-dose versions for younger children to try to reduce vaccine side effects.
How effective are the vaccines?
Moderna's vaccine was estimated to be 50.6 per cent effective at preventing symptomatic infections among children six months to under two years old and 36.8 per cent effective at doing so in children aged two to six in a clinical trial of more than 5,000 subjects.
It is not yet known how effective the Pfizer-BioNTech vaccine is at preventing infection in people under five because of the low number of symptomatic Covid-19 cases among the children in its trial. An early analysis based on 10 symptomatic cases suggested a vaccine efficacy of 80.3 per cent in this group. Once 21 children in the trial contract symptomatic Covid-19, the companies can finalise the vaccine's efficacy.
Both the vaccines' trials showed that they generated a similar immune response to those seen in older age groups.
Are the vaccines safe for children?
Neither vaccine has raised any serious safety issues in the trials.
Heart inflammation known as myocarditis and pericarditis has been found to be a rare side effect of the vaccines in young men, but there have been few cases reported in the United States in children aged five to 11, and none in either of these trials for the youngest age group.
What are the side effects?
Some children reported pain and swelling at the injection site after vaccination from both vaccines.
Pfizer-BioNTech's low-dose vaccine was generally better tolerated, with only slightly more participants who received the vaccine complaining of fever, irritability or fatigue when compared with those who received a placebo. In ages two to four, 33.7 per cent of participants who received their second dose of the vaccine reported one of these side effects, compared with 32.2 per cent who received a placebo.
For Moderna's vaccine, 58.9 per cent of three- to five-year-olds reported some type of reaction like fever, headache or fatigue after receiving their second dose, compared with 37.2 per cent of participants who received a placebo.
Why vaccinate young children?
Covid-19 is generally milder in children than adults, but there have been more than 440 US Covid-19 deaths among those under five, according to FDA officials.
Data has shown that in older children and adults, vaccines protect against hospitalisation and death, according to Dr Matthew Harris, a paediatric emergency medicine physician at Northwell Health in New York.
People who had a Covid-19 infection and received a booster shot had the most protection against Covid-19, he said.


