SGX queries Biolidics on timing, details of A*Star licence announcement
Sign up now: Get ST's newsletters delivered to your inbox
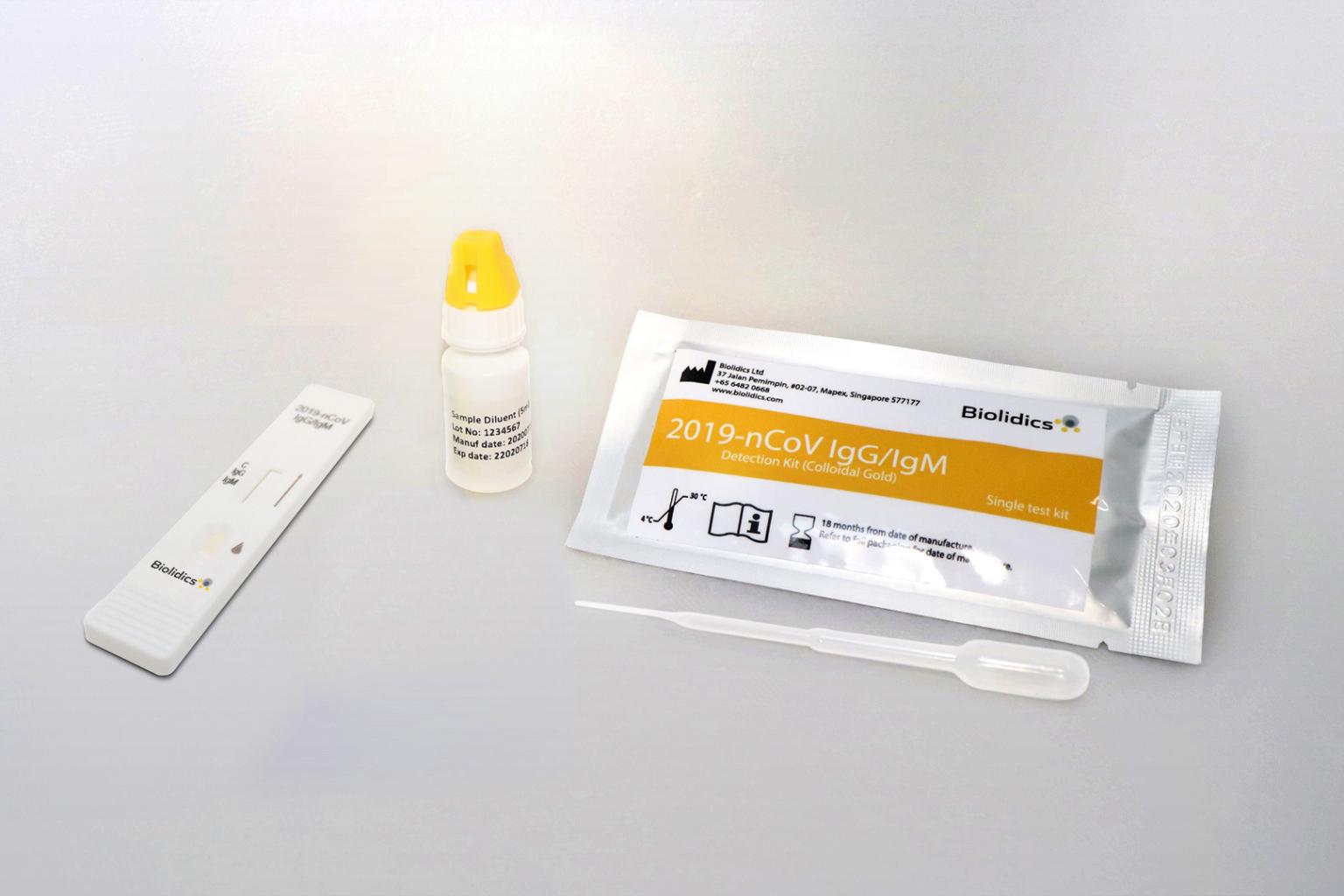
Biolidics' rapid test kit for Covid-19.
PHOTO: BIOLIDICS LIMITED
SINGAPORE (THE BUSINESS TIMES) - The Singapore Exchange (SGX) on Wednesday (July 15) posed a slate of queries to Biolidics about the details of a licence agreement the Catalist-listed firm had inked as well as the timing of its trading halt and announcement.
In a filing on Tuesday night, Biolidics said it had on July 9 entered into a five-year licence agreement with Accelerate Technologies, the commercialisation arm of the Agency for Science, Technology and Research (A*Star), to use the agency's technology for the detection of Covid-19 viral spike/Angiotensin Converting Enzyme 2 (ACE2) blocking antibodies.
Biolidics added that it was aiming to develop new serology tests which incorporate the technology.
Shares of the Covid-19 test kit maker jumped 29.3 per cent on Monday before it called for a trading halt at 12.19pm. It then released the A*Star announcement on Tuesday at 10.46pm, before lifting the halt on Wednesday morning ahead of the market's opening bell. The shares fell heavily, closing down 8.5 cents or 16.4 per cent, at 43.5 cents.
SGX asked Biolidics to provide reasons for the delay in announcing the agreement, seeing as it was signed several days ago, and whether its sponsor was aware and consulted on the disclosure of the matter, and what advice the sponsor gave.
The bourse operator also asked when Biolidics' board and sponsor had approved the announcement.
Under the five-year licence pact, Accelerate Technologies is granting Biolidics non-exclusive worldwide rights to the technology for diagnostic use, to further develop the technology and to market new serology tests that incorporate the technology.
SGX asked Biolidics whether it provided any consideration or outlay for being granted these rights to use A*Star's technology.
Biolidics is also required to disclose the rights assignment for the products and technologies that it will subsequently develop using the technology. "Are there any differences between how these rights are assigned during and after the (five-year) term? Does Biolidics have exclusive rights to the said products or technologies?" SGX added.
As for Biolidics' intention to further develop the technology, the bourse operator questioned the company on its ability and efforts to do so. SGX requested the projected timeline or milestone, if any, for Biolidics to successfully develop the technology.
SGX also asked whether there were conditions to be met - such as the company having to complete the development of the technology - before Biolidics can market the serology tests that incorporate the technology, into its Covid-19 antibody test kit or into any other form of serology tests.
Lastly, Biolidics is required to disclose when it expects to be able to market the serology tests that incorporate the technology, as well as how far along the company is in successfully incorporating the technology into its Covid-19 antibody test kit or any other form of serology tests.
Biolidics must release its responses to these questions via a filing on SGXNet by Thursday.
Shares of Biolidics have swung in the last few months amid a series of rapid-fire announcements.
Here is a timeline of recent events:
March 30: Biolidics announces it is launching its Covid-19 rapid test kit by end-April.
April 1: It obtains authorisation for the use of test kits in the Philippines.
April 6: Biolidics' test kits are allowed to be sold in the European Union.
April 13: Biolidics announces plans to distribute its test kits in the United States.
April 20: Biolidics gets permission to market and sell its test kits in the US.
April 23: Biolidics signs exclusive US distribution deal with Aytu Bioscience for its test kits.
April 26: Biolidics responds to Singapore Exchange (SGX) queries over its appointment of scandal-hit Aytu that it does not expect the agreement to have a material impact on its business plans.
May 8: Biolidics appoints Mediproud as the non-exclusive distributor of its test kits in Thailand.
May 11: Biolidics appoints Clearbridge Health as non-exclusive regional distributor of its test kits in the Philippines, Indonesia, Myanmar and Vietnam.
June 27: Biolidics calls off the sale of its test kits in the US, citing increased competition. It announces it is working with Aytu to develop a new test kit with broader use outside of laboratory or clinical settings.
July 3: SGX queries Biolidics on its scrapped distribution deal with Aytu.
July 8: Biolidics signs five-year agreement with China's LC-Bio to develop cancer-diagnostic solutions.
July 14: Biolidics signs five-year licence agreement with Accelerate Technologies for the use of Covid-19 viral spike detection technology. It announces plans to develop new tests incorporating the technology.


