Biolidics calls off US sale of Covid-19 test kits, to develop new kit with broader use
Sign up now: Get ST's newsletters delivered to your inbox
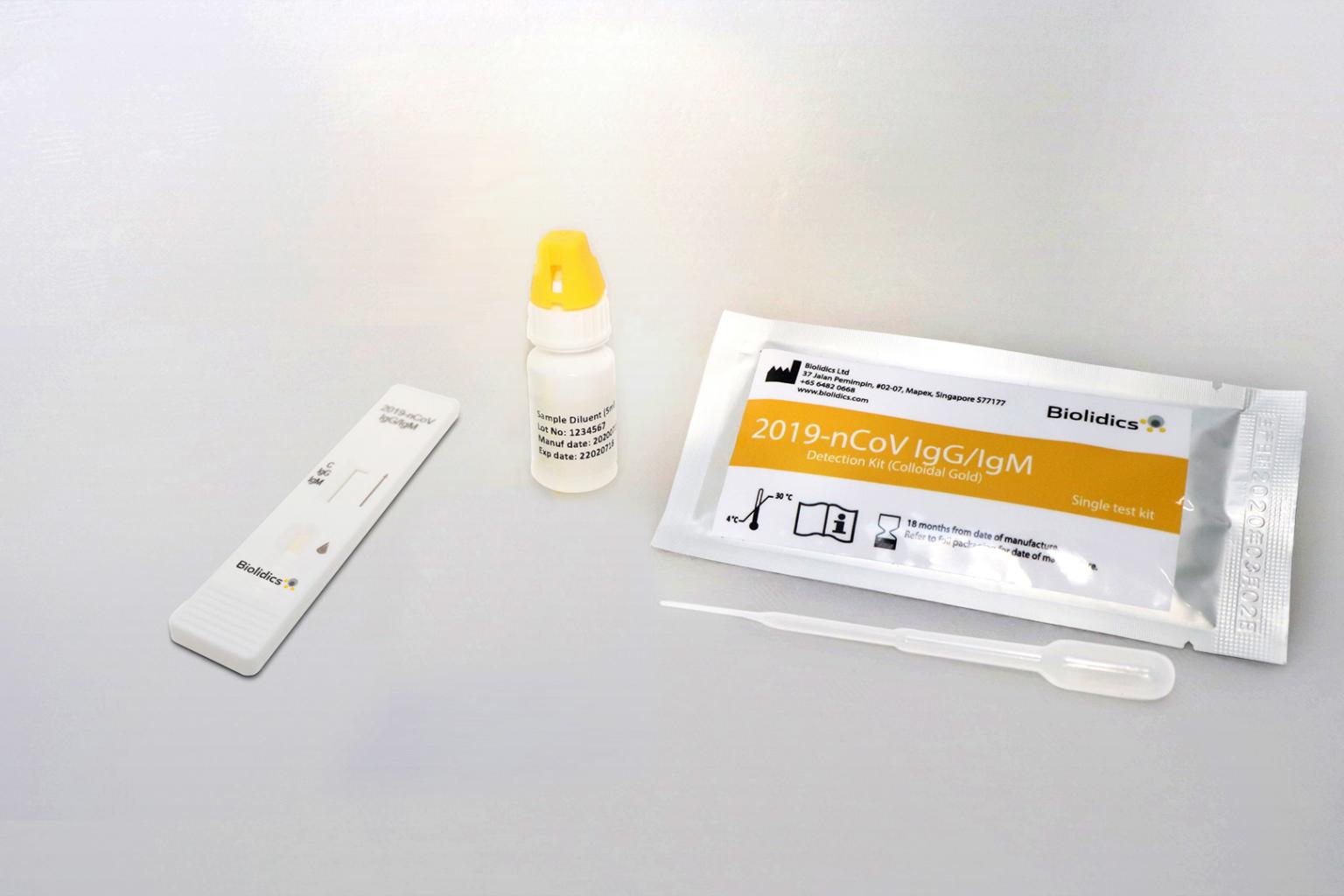
Biolidics intends to continue to distribute, market and/or sell its current test kits in markets outside the US.
PHOTO: BIOLIDICS LIMITED
SINGAPORE (THE BUSINESS TIMES) - Biolidics has signed a non-binding letter of intent (LOI) with Aytu Bioscience to jointly develop a new test kit with broader use outside of laboratory or clinical settings.
This comes after both companies had mutually agreed to terminate an earlier distribution agreement for Biolidics' Covid-19 antibody test kits in the US amid increased competition for such kits, Biolidics said on Sunday (June 28).
With the distribution deal scrapped, Biolidics will need to refund Aytu in full for all deposits paid with respect to undelivered orders of its test kits.
The Catalist-listed company also intends to apply to voluntarily withdraw its application to the US Food and Drug Administration (FDA) for Emergency Use Authorisation (EUA) pursuant to the FDA's serology test policy.
As a consequence, its current test kits will no longer be available in the US market, it said.
However, Biolidics intends to continue to distribute, market and/or sell its current test kits in markets outside the US. It has signed distribution agreements for the sale of its kits in Thailand, the Philippines, Indonesia, Myanmar, Vietnam, and Hong Kong.
Earlier in April, Biolidics signed a one-year agreement with Aytu for the distribution of its test kits in the US after completing its US FDA listing.
In April, Biolidics had assessed that the distribution agreement with Aytu would likely have contributed positively to its revenue for the current financial year ending Dec 31, 2020. However, revenue from US sales of the test kits under the agreement to date was not material, it said on Sunday.
The company's existing test kits are limited to testing in laboratories, or by healthcare workers at the point-of-care.
It now plans to refocus efforts and resources into the proposed project with Aytu to develop a new serology test kit with broader use and applications outside the laboratory or clinical settings - for example, by individuals at home - in the US, which Biolidics believes may present a better commercial opportunity.
"The company understands that there is currently no EUA authorised serology tests which could be used outside of the laboratory or clinical settings in the US market," Biolidics said.
Both parties are negotiating the terms of a binding definitive agreement for the proposed project for the new test kit.
The termination of the distribution deal and the signing of the LOI are not expected to have a material impact on the group's earnings per share and net tangible assets per share for the current financial year, Biolidics said.
The company noted that results from all serology test kits are not to be used for confirmatory testing or as the sole basis for diagnosis. They will have to be interpreted together with clinical presentation and are to be confirmed with supplementary testing.
Its shares were trading at 33.5 cents as at 9.45am on Monday, down 7.5 cents or 18.3 per cent.


