US FDA mandates breast density information with mammography results
Sign up now: Get ST's newsletters delivered to your inbox
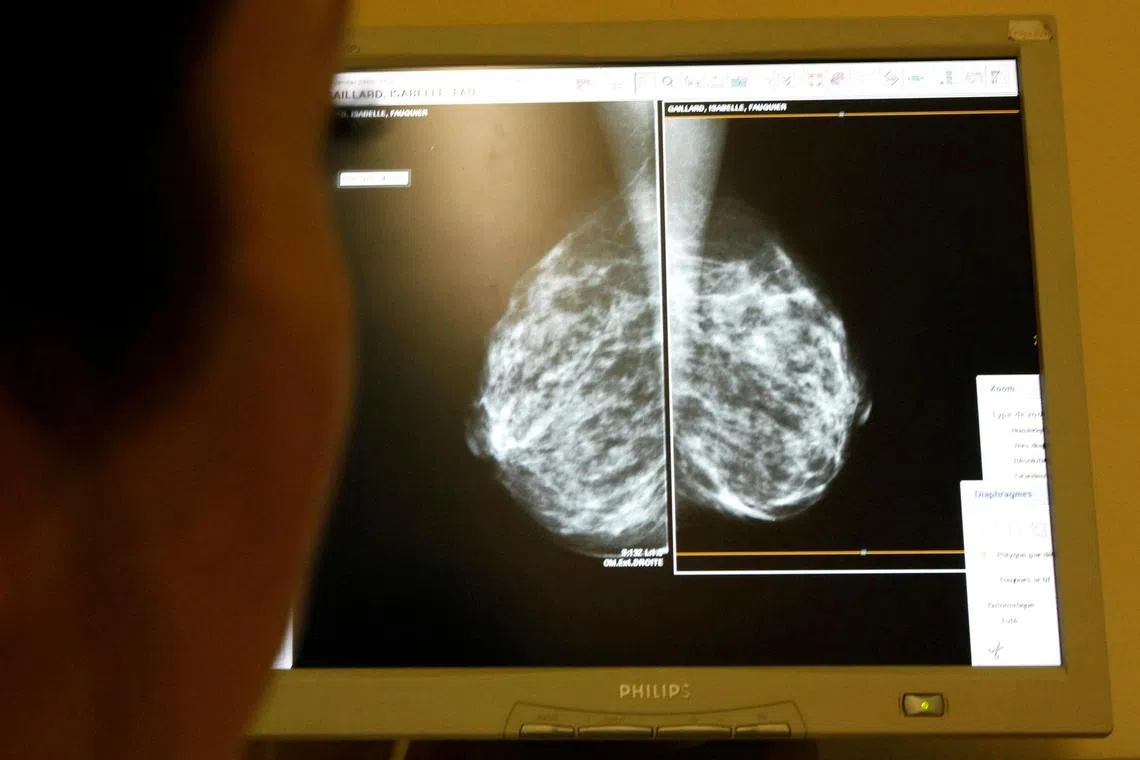
The new rules will help to better advise patients on the potential need for further evaluation or a repeat mammogram.
PHOTO: REUTERS
MARYLAND - The United States Food and Drug Administration said on Thursday that all mammography facilities in the country will be required to notify patients about the density of their breast tissues, as it could lead to earlier detection of tumours.
Breast density can influence the accuracy of mammography, which relies on X-rays passing through breast tissues to diagnose and locate tumors.
Mammograms remain the best tool for screening and detection of breast cancer, which is the second most common form of cancer among women in the US, according to the Centres for Disease Control and Prevention.
The agency recommends patients with dense breasts – a normal and common finding in mammograms – to discuss their individual situation with their respective healthcare providers.
The updated rules will help ensure patients receive information on the potential need for further evaluation or a repeat mammogram, the health regulator said.
“Increasing early detection is the best way to improve outcomes, and this new standard does just that,” Congressman Brian Fitzpatrick said in a statement. REUTERS


