Pfizer starts shipping vaccine to UK from Belgium
Sign up now: Get ST's newsletters delivered to your inbox

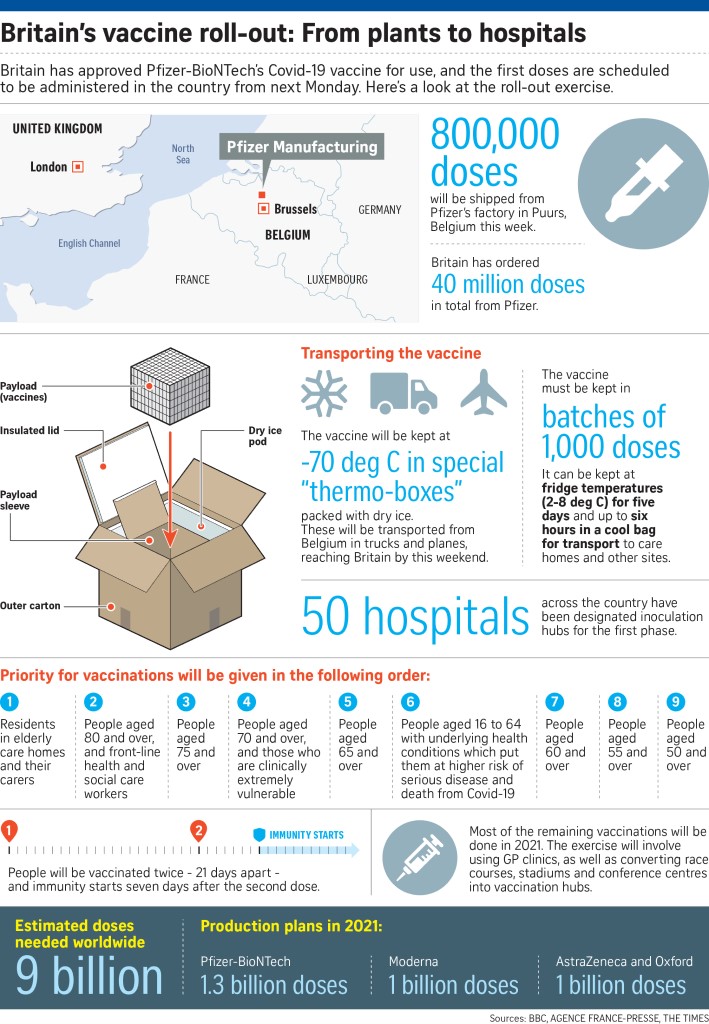
A temperature-controlled truck leaving the Pfizer production plant in Puurs, Belgium, yesterday. The US pharmaceutical giant plans to ship an initial 800,000 doses to Britain in the coming days from its Puurs facility so that they will be ready to be administered from Monday.
PHOTO: BLOOMBERG
BRUSSELS • Doses of the vaccine made by US pharmaceutical giant Pfizer and a small German company BioNTech, the first rigorously tested coronavirus vaccine given the green light for use, are being packaged in Belgium for shipment to Britain this week.
Pfizer plans to ship an initial 800,000 doses to Britain in the coming days from its production plant in Puurs, Belgium, so that they will be ready to be administered from Monday.
Britain on Wednesday gave emergency use authorisation to the Covid-19 vaccine, becoming the first Western country to pave the way for mass inoculations against a disease that has killed more than 1.4 million people worldwide.
Within weeks, more government approvals for the Pfizer-BioNTech and rival Moderna vaccines are expected as vaccine makers get ready to start shipping millions of doses.
BioNTech said the first doses were being prepared for ship-ment from Tuesday night. Boxes of several thousand doses each are packaged with dry ice to keep them at the South Pole-like temperatures they require, before they are put on trucks or planes and taken to Britain.
British hospitals have already begun e-mailing staff to schedule vaccinations, with a London hos-pital system indicating that its first doses will be given at 7am on Monday.
Britain placed advance orders for 40 million doses of the Pfizer vaccine, but most of these are expected to be administered next year. Each patient needs two doses, spaced three weeks apart.
The much-awaited vaccine comes as England emerged from a month-long national lockdown on Wednesday, replaced by a three-tiered system of restrictions, with about 40 per cent of people subjected to the toughest tier three.
Under the new system, shops will be allowed to reopen across England, giving a Christmas bonanza to the struggling retail sector, but all bars and restaurants in tier three areas will remain closed.
On Wednesday, the country recorded 16,170 new coronavirus cases for a total of 1,659,256. Covid-19 related deaths rose by 648 to 59,699, government data showed.
Expectations are growing that more vaccine approvals will be granted within weeks.
The European Medicines Agency said it is expected to make a decision on Pfizer-BioNTech's vaccine "by Dec 29 at the latest", while a ruling on Moderna's version should follow by Jan 12. The European Commission in Brussels needs to give its final approval.
Pfizer-BioNTech and Moderna have requested emergency use authorisation for their vaccines from the United States Food and Drug Administration (FDA).
The FDA carries out its own analysis of the vaccine and calls on an independent advisory committee.
The advisory committee is scheduled for a Dec 10 meeting on the Pfizer-BioNTech vaccine, and for Moderna on Dec 17. FDA decisions on the two drugs should follow those meetings.
If the agency approves them, the vaccines could be available in the US this month.
In Singapore, the Health Sciences Authority has started eva-luating the available data submitted by Moderna, which is seeking approval for use of its vaccine in the Republic.
Australia's pharmaceutical regulator said yesterday it is on course to review the Pfizer-BioNTech vaccine by next month, with the country sticking to a March timetable to start vaccinations.
Russia has already begun testing its Sputnik V vaccine on a large number of volunteers, and President Vladimir Putin has told the authorities to begin large-scale vaccinations among at-risk populations from next week.
NYTIMES, AGENCE FRANCE-PRESSE, REUTERS
SEE WORLD


