Pfizer asks UK regulator to approve Covid-19 vaccine for teenagers
Sign up now: Get ST's newsletters delivered to your inbox
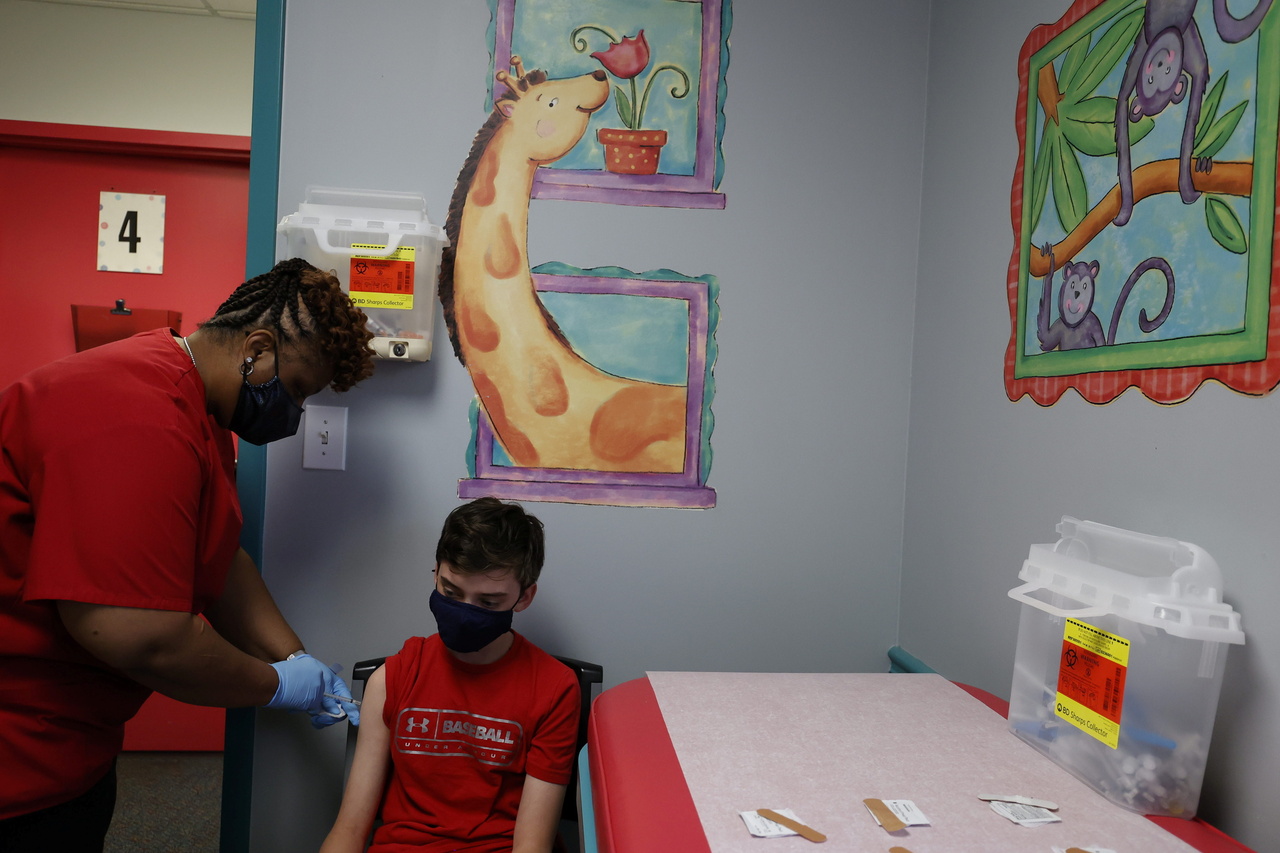
In March, Pfizer and BioNTech said their Covid-19 vaccine was safe, effective and produced robust antibody responses in 12- to 15-year olds.
PHOTO: REUTERS
LONDON (REUTERS) - Pfizer has submitted data on its Covid-19 vaccine among 12- to 15-year olds to Britain's health regulator, a company spokesman said on Tuesday (May 11), paving the way for its authorisation to inoculate children in the country.
"I can confirm we've submitted data on the efficacy of the vaccine in 12 to 15 years olds for review by UK MHRA," the spokesman told Reuters by email.
The move comes a day after US regulators authorised the vaccine made by Pfizer and BioNTech for use in children as young as 12.
In March, Pfizer and BioNTech said their Covid-19 vaccine was safe, effective and produced robust antibody responses in 12- to 15-year olds, citing a trial of 2,260 adolescents, which resulted in 100 per cent efficacy in preventing the illness.
Pfizer has said it expects to have safety and efficacy data on the vaccine for children aged 2 to 11 in September, when it plans to ask for that age group to be included in the emergency authorisation.
The Telegraph earlier reported that Pfizer had asked Britain's Medicines and Healthcare products Regulatory Agency for approval for the vaccine.


