HSA warns against buying and consuming juice drinks from Malaysia that contain steroids
Sign up now: Get ST's newsletters delivered to your inbox

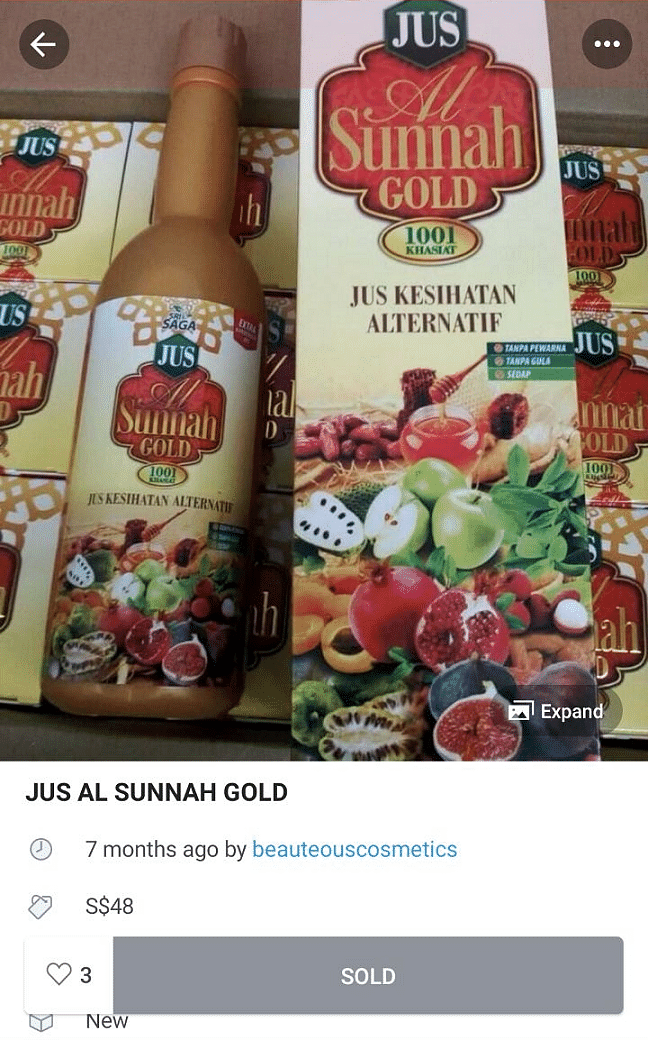
Jus Al Sunnah Gold 1001 Khasiat Jus Alternatif and Jus Al Sunnah 1001 Khasiat Jus Alternatif were tested and found to contain potent medicinal ingredients.
PHOTOS: HEALTH SCIENCES AUTHORITY
SINGAPORE - The Health Sciences Authority (HSA) has warned the public to avoid buying and consuming two juice drink products from Malaysia that have caused serious steroid-induced side effects in at least five people, including a child.
HSA said on Thursday (Sept 27) that the two products, Jus Al Sunnah Gold 1001 Khasiat Jus Alternatif and Jus Al Sunnah 1001 Khasiat Jus Alternatif, were tested and found to contain potent medicinal ingredients.
The undeclared ingredients included dexamethasone and prednisolone, which are potent steroids usually prescribed for inflammatory conditions that should only be taken under strict medical supervision.
Long-term unsupervised use of oral steroids can cause Cushing's syndrome, diabetes and high blood pressure, HSA said. Users may also be susceptible to bone disorders and an increased risk of infections and muscle weakness.
Diclofenac, a potent painkiller, was also found in the "Gold" variant of the product. It can cause serious gastric bleeding, heart attacks and stroke when used over a prolonged period, especially in patients with underlying heart conditions, the authority said.
Last Saturday (Sept 22), Chinese evening daily Shin Min Daily News reported that a 28-year-old woman had died in hospital after consuming a juice drink that contained dexamethasone for about six months.
In response to queries, the Singapore Civil Defence Force said that it responded to a call for assistance at 428 Yishun Avenue 11 at around 1.30pm on Sept 21. A Malay woman in her 20s was taken to Khoo Teck Puat Hospital.
The Straits Times understands that her death has been classified as a coroner's case and her cause of death has yet to be determined by a coroner's inquiry.
The woman, Ms Titinlismawati Sajali, had been taking the drink for pain relief. Her family told Shin Min that she had bought the product in the Geylang Serai area.
Checks by ST also found that Jus Al Sunnah products were available on online retail platform Carousell and were advertised as a treatment for various conditions, including body pain, tiredness and gout. The listings have since been removed.
In June, Brunei's Ministry of Health issued a statement banning the import and sale of Jus Al Sunnah products in the country after their tests detected dexamethasone.
On Tuesday, Malaysia's Health Ministry director-general Noor Hisham Abdullah also issued a ban on the product. He said that the company listed as the product's producer, Sri Saga Marketing S/B, did not exist.
HSA advised members of the public to consult a doctor if they are taking the products as sudden discontinuation of steroids without medical supervision can cause withdrawal symptoms such as fatigue, confusion and low blood pressure.
The authority also said that sellers must stop selling the products immediately.


