Covid-19 breathalysers could be used on large scale in Singapore soon
Sign up now: Get ST's newsletters delivered to your inbox
SINGAPORE - Mass deployment of Covid-19 breathalysers that produce results on the spot may soon happen here. This will facilitate safe travel arrangements and screenings at large-scale events.
The Straits Times has learnt that local medtech firm Silver Factory Technology is working with the National Centre for Infectious Diseases (NCID), Changi Airport and security firm Certis to develop the breathalysers.
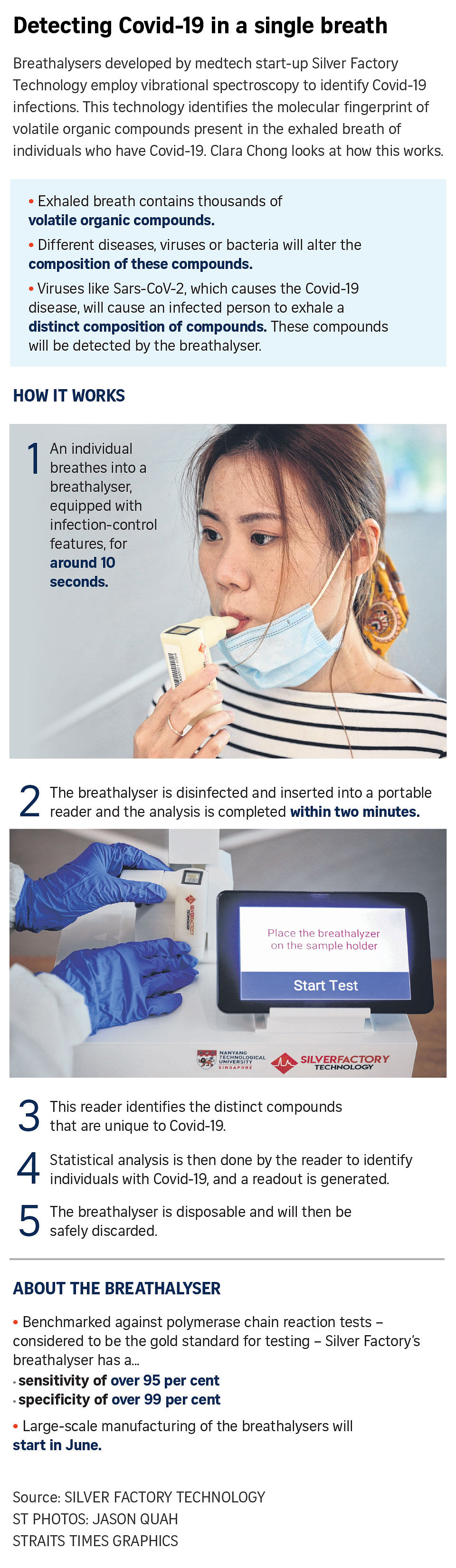
Silver Factory's breathalyser, TracieX, has been shown to be almost as accurate as polymerase chain reaction (PCR) tests, while taking a fraction of the time - at two minutes - to turn in results.
Compared with PCR tests - the gold standard for testing - the disposable breathalyser's sensitivity rate so far has been over 95 per cent, and its specificity, more than 99 per cent.
It is also cheaper, costing around US$20 (S$26) each currently.
Trials have already started in NCID, Changi Airport Terminal 1 and Certis, with more than 400 individuals tested to date, Silver Factory said.
In an unblinded trial (where participants have prior knowledge of their Covid-19 status) at Changi that began in March, arriving passengers were selected randomly and asked to volunteer for the trial by providing a breath sample for analysis.
The test site was set up with the help of NCID, which evaluated its biosafety protocols and monitored and analysed the data collected.
"Breathalyser tests are less invasive, more cost-effective and can deliver results in two or fewer minutes, which if proven reliable, will make the airport experience for passengers a smoother one and give the reopening efforts a boost," said Mr Albert Lim, senior vice-president of passenger experience in the Changi Airport Group.
Silver Factory, founded last January as a spin-off from Nanyang Technological University, aims to produce at least 200,000 breathalysers a month from June this year, potentially ramping up production to two million a month.
The product's cost price is expected to fall once large-scale production commences.
Sensor chips will be produced in its upcoming 300 sq m pilot plant in Tuas - roughly the size of three five-room Housing Board flats - while home-grown manufacturing company Dou Yee Technologies will assemble the devices.
Further studies are also set to begin soon in Changi, NCID and some hospitals in Malaysia, Silver Factory said.
At least 1,000 participants will be recruited in Singapore in all for both the current and upcoming studies, with this number potentially extending to 2,000, said Dr Shawn Vasoo, head of the Infectious Disease Research Laboratory and clinical director in NCID.
Associate Professor Ling Xing Yi, one of the co-founders of Silver Factory, told ST that trials will be expanded to Malaysia due to the low number of Covid-19-positive cases here.
Silver Factory aims to apply for regulatory approval from the Health Sciences Authority (HSA) by June this year.
Once these breathalysers are approved, Certis will also use them in its current Covid-19 testing operations, Mr Joseph Tan, the security firm's senior managing director and head of technology services, told ST.
The breathalyser "delivers results in just two minutes with a high degree of accuracy and is able to do crowd screening... for aviation and cruise operations, government checkpoints, immigration clearance and at healthcare and medical facilities," Mr Tan added.
Certis will have exclusive rights to market this breathalyser in its key markets of Singapore, Australia, Hong Kong, Macau and Qatar.
Another start-up, Breathonix, is currently running trials at three sites - Changi Airport, NCID and in Dubai - with more than 3,000 people tested in total.
It aims to get HSA approval by the first half of this year.
"This trial will help Breathonix better understand and improve the actual deployment workflow for our breath test station," said Dr Jia Zhunan, chief executive of Breathonix.

Start-up Breathonix is currently running trials at three sites with over 3,000 people tested in total.
ST PHOTO: TIMOTHY DAVID


