Singapore's expert panel on Covid-19 vaccination continues to recommend Pfizer, Moderna
Sign up now: Get ST's newsletters delivered to your inbox
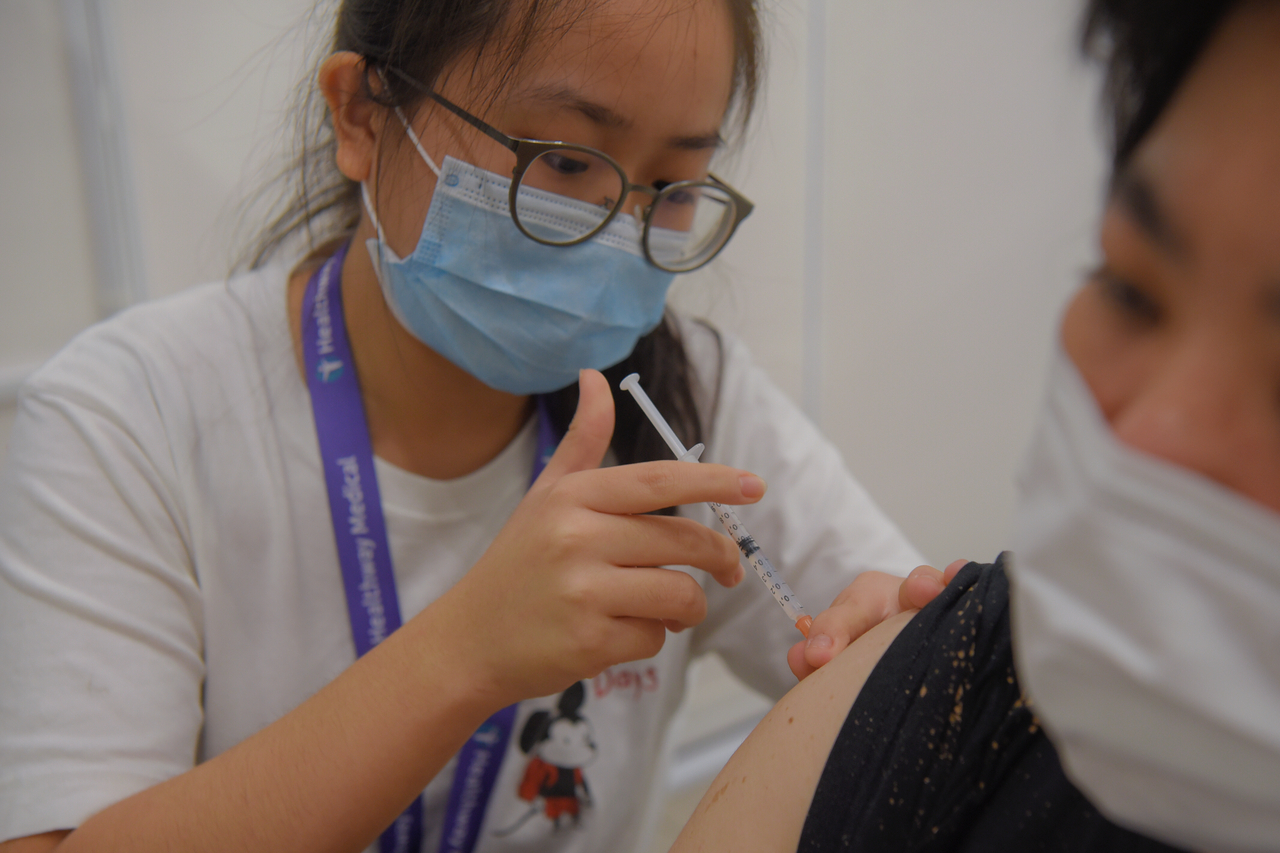
The expert committee said the Pfizer-BioNTech and Moderna mRNA vaccines, have been consistently shown to be highly efficacious, with an efficacy rate of around 90 per cent.
ST PHOTO: MARK CHEONG
SINGAPORE - The expert committee on Covid-19 vaccination continues to endorse the effectiveness of the mRNA-based Pfizer-BioNTech and Moderna vaccines used in Singapore, it said on Monday (June 7).
The committee said it is aware of messages that are being disseminated on social media that claim that mRNA-based vaccines are ineffective against new variants of concern, and that vaccines that use an inactivated virus would provide superior protection.
The China-produced Sinovac shot is one example of a vaccine that uses inactivated viruses.
"Our assessment, based on a continual review of data and evidence, remains that the mRNA vaccines are safe and highly effective, and continue to show protection against the variants of concern," the committee said in a statement.
"The inactivated virus Covid-19 vaccines have variable protection and there is currently no evidence to suggest that inactivated virus vaccines demonstrate higher vaccine efficacy against variants of concern than the mRNA vaccines."
Covid-19 variants of concern include those like the more infectious B16172 or "delta" variant that has played a role in the current outbreak in Singapore.
The expert committee said the Pfizer-BioNTech and Moderna mRNA vaccines, which are currently the only two available under Singapore's national vaccination programme, have consistently shown to be highly efficacious, with an efficacy rate of around 90 per cent.
They are especially effective in protecting against severe Covid-19 disease and hospitalisation, the committee added.
"This was first demonstrated in the pivotal Phase 3 clinical trials, and further supported with the data from actual roll-outs in the real-world, including in the United States, Britain and Israel," the committee said.
"This includes protection against key variants of concern, such as the alpha and beta variants, which were the predominant strains circulating in these countries."
The committee further said that despite concerns over the delta variant, emerging data shows that the mRNA vaccines continue to be effective.
It cited a study in Britain which demonstrated that two doses of the Pfizer-BioNTech mRNA vaccine conferred about 88 per cent protection against symptomatic Covid-19, even with the delta variant.
"While further studies are required before a definitive conclusion can be made, the available data globally indicates that substantial protection is preserved."
The committee noted that no vaccine provides 100 per cent protection, and with the emergence and spread of new variants due to mutations, infections can occur despite vaccination.
But it stressed that the detection of asymptomatic to mild infections among patients with the delta variant does not indicate a lack of protection.
This is in keeping with the global evidence that the mRNA vaccines have a high level of protection against symptomatic and severe disease, it added.
The Pfizer-BioNTech and Moderna vaccines have also been approved by multiple reputable international regulatory bodies.
The manufacturers have publicly released their detailed study protocols and openly published their findings to be scrutinised by the scientific community after peer review.
Detailed assessments of these vaccines by regulatory bodies, such as the Food and Drug Administration in the United States and the British Medicines and Healthcare Products Regulatory Agency, have also been published.
On the other hand, the committee said the Sinovac shot has yet to meet the requirements for authorisation by Singapore's Health Sciences Authority, as additional safety and quality data required to meet the standards of the evaluation are still pending.
"The Sinovac vaccine has also shown variable protection across multiple studies carried out internationally," the committee said.
"The most complete analysis of the vaccine showed a vaccine efficacy of 51 per cent."
It also said the protection Sinovac provides against newer variants, such as the delta variant, and under real-world conditions remains unknown.
The Sinovac vaccine will soon be available in Singapore, unsubsidised, at private healthcare providers under the Special Access Route.
It has been qualified by the World Health Organisation for emergency use in those aged 18 and above.
But this means it is not an option for children and adolescents under 18 globally, nor in Singapore, the committee said in its statement.
"It is critical that medical professionals do not spread unsubstantiated and unscientific information," the committee added.
"The public has a right to expect medical professionals to give advice based on facts and not on unproven assertions. The public should rely on reputable sources of scientific and medical information, and verify opinions shared by others against these."
In a separate Facebook post on Monday, Associate Professor David Lye, a senior infectious diseases specialist, also spoke up against misinformation being spread about Covid-19 vaccines by doctors, among others.


