Rapid Covid-19 test kits not used in Singapore as they can miss true cases
Sign up now: Get ST's newsletters delivered to your inbox
Test kits that can determine if a person has Covid-19 in minutes are not being used in national swabbing exercises as they are not sensitive enough to detect every infection.
"They give false negative results - they miss true cases," said Associate Professor Raymond Lin, director of the National Public Health Laboratory at the National Centre for Infectious Diseases (NCID).
Prof Lin told The Straits Times: "Currently available antigen rapid test kits can miss as many as one in six cases, or even more. If we miss cases, we will have more outbreaks."
NCID will continue to look for test kits that are fast and accurate, he added.
More than 120 Covid-19 diagnostic test kits have received provisional authorisation from the Health Sciences Authority (HSA), ranging from polymerase chain reaction (PCR) kits to antigen and serology ones.
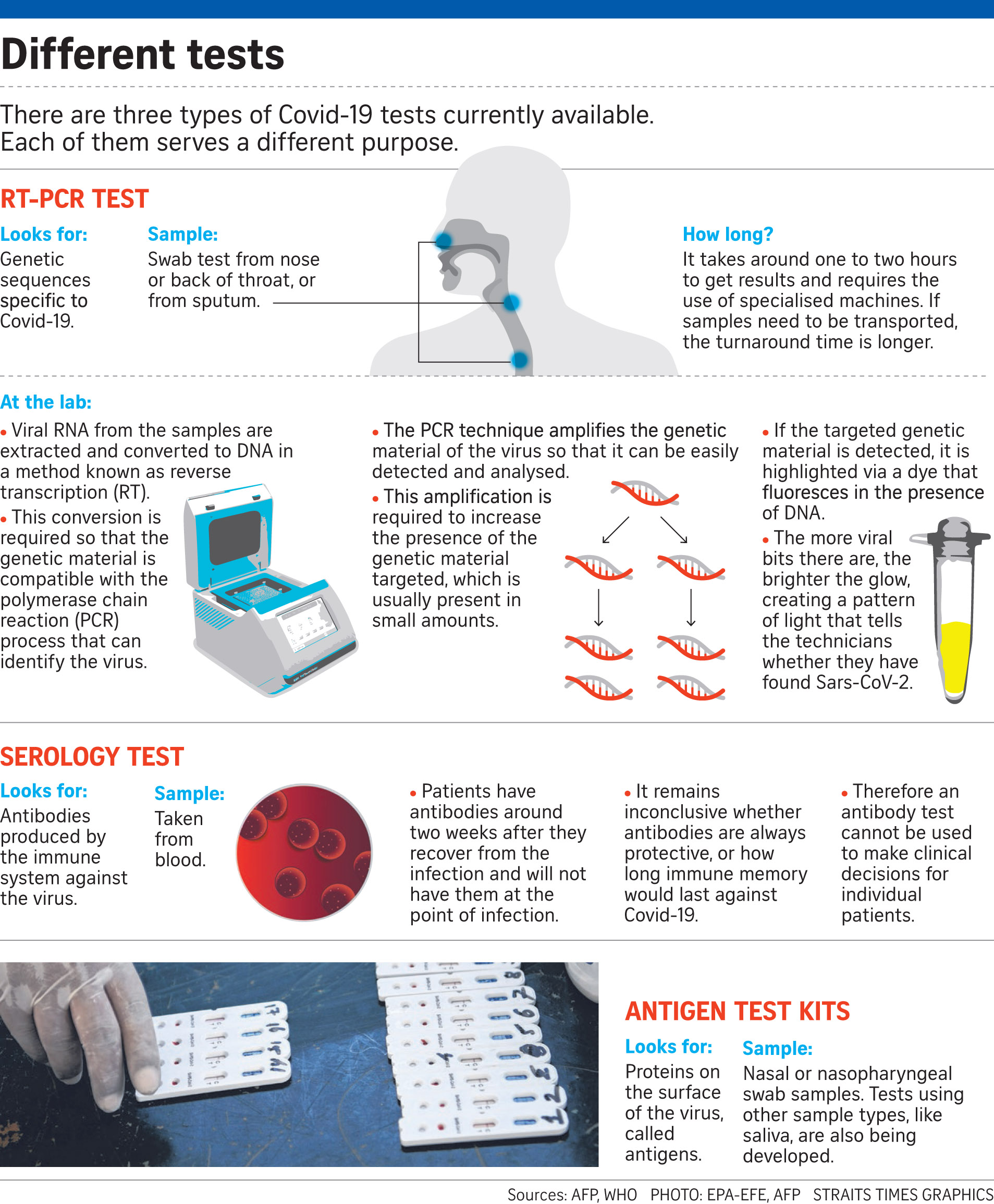
All three work differently.
PCR tests look for fragments of the virus' genetic material in a patient sample and are considered the "gold standard" test for Covid-19.
But the entire process - from swabbing a patient to sending the sample to a laboratory and the PCR process itself - can take up to a day. It can also be costly, and requires trained personnel and specialised equipment to administer.
Antigen test kits, on the other hand, are point-of-care tests that look for proteins on the virus surface and can yield quick results, usually within minutes.
Serology tests, which detect the presence of antibodies, are mainly used to tell if a person has been infected in the past, and not to diagnose patients.
Last Friday, the World Health Organisation set out its general recommendations on the use of such rapid test kits.
The guidelines include specific circumstances, such as dealing with suspected outbreaks of Covid-19 in remote settings where PCR testing is not immediately available, and for monitoring trends in disease incidence in communities.
Professor Teo Yik-Ying, dean of the National University of Singapore's Saw Swee Hock School of Public Health, noted that because rapid diagnostic tests are cheaper and less invasive than PCR tests, their prudent and repeated deployment could supplement national measures to control Covid-19.
But they have to be used in a way to reduce the potential of false readings - whether positive or negative - from hampering the system, he said.
If protocol dictates that readings from rapid tests are acted upon, for example, false negatives could seed new outbreaks, while false positives could add strain on the hospital system and the patient's pockets.
But Prof Teo said there was potential for rapid test kits to be deployed in certain scenarios, such as in population segments undergoing routine monitoring.
"Migrant workers are being tested fortnightly, and the process of undergoing PCR swabs every 14 days can actually be quite traumatising since the nose swab is pretty unpleasant," said Prof Teo.
It could be possible to use rapid tests, perhaps two to three times every fortnight, to perform the same surveillance, he noted.
"Because the cost of some of these rapid tests is lower, a higher frequency of testing may still be less costly than the PCR swab test - and certainly, if there is sufficient demand, the price will also drop."
American medical technology company Becton Dickinson (BD) has developed a rapid test for Covid-19 that has obtained provisional approval from HSA.
Its BD Veritor Sars-CoV-2 Rapid Antigen test can detect an infection within 15 minutes, said its spokesman, who added that every test has its pros and cons.
PCR tests are costly, require skilled molecular technicians and take a longer time to yield results. Their robustness also depends on the segment of the viral genetic material being flagged.
Rapid tests can complement PCR tests, not replace them, as different tests are appropriate for use at different times, the spokesman said.
"Having rapid antigen tests at point-of-care settings... complements PCR testing, allows hospitals and laboratories to prioritise their PCR tests for urgent cases and not overburden the laboratory system," he added.


