HSA probing unauthorised distribution, supply of SG Diagnostics ART kits
Sign up now: Get ST's newsletters delivered to your inbox
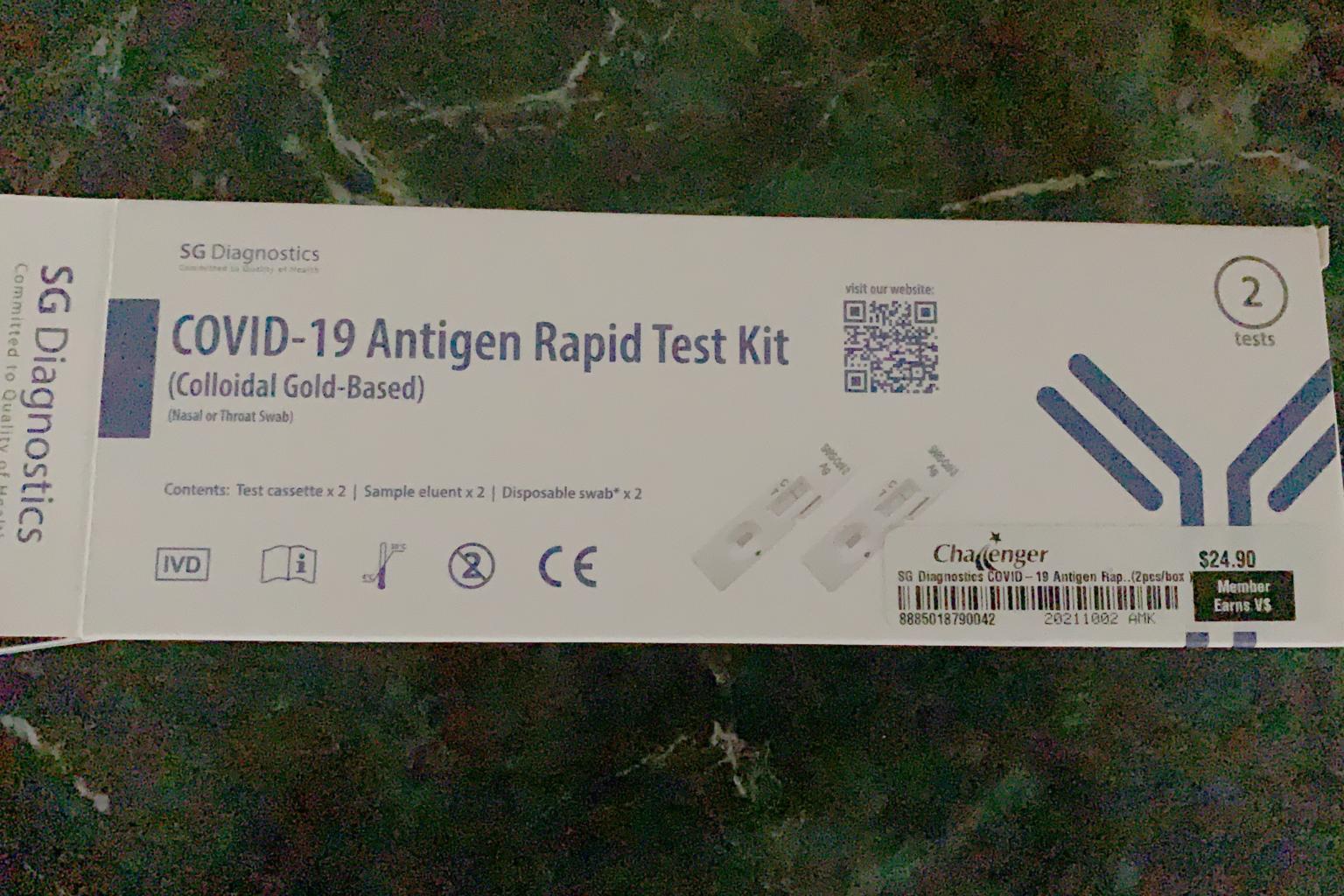
A photo of an SG Diagnostics ART kit that was purchased in Challenger for $24.90.
PHOTO: ST READER
SINGAPORE - The Health Sciences Authority (HSA) is investigating the unauthorised distribution and supply of antigen rapid test (ART) kits manufactured by Singapore-based company SG Diagnostics.
They are meant for professional use in "healthcare settings by healthcare professionals who are trained to administer the test and validate the results", said the authority on Thursday (Oct 14) in response to queries from The Straits Times.
The kits should not be used by untrained lay users, who may derive inaccurate results, the HSA said, adding that this could lead to them not getting timely treatment if they have Covid-19, or failing to take necessary precautions to protect themselves and the people around them.
SG Diagnostics had urged consumers in an advertisement in ST Classifieds on Tuesday to stop using the product.
When ST called the SG Diagnostics hotline listed in the ad, an operator said buyers are advised to return the ART kits to their suppliers and ask for a refund.
ST has contacted SG Diagnostics for comment.
Sellers had put up listings on e-commerce platforms like Shopee, Lazada and Carousell, pricing a box of two kits at about $18.
Listings for group orders were also seen on shopping platform WeBuy.
Many of the listings have been taken down.
The kits were also seen to be for sale in physical stores like IT retailer Challenger.
The HSA said regulatory and enforcement actions under the Health Products Act will be taken against any party involved in unauthorised distribution and supply of the kits.
It advised consumers to purchase and use self-administered ART kits approved in Singapore.
The list of kits can be found on the HSA's website.
For inquiries on returning the kits, consumers should call the SG Diagnostics hotline on 8030-5766 or e-mail info@sgdiagnostics.com


