Duke-NUS to co-develop, produce one-of-its-kind test kit with partners
New antibodies test, known as cPass, is available to hospitals in S'pore
Sign up now: Get ST's newsletters delivered to your inbox
The cPass test kit can detect antibodies capable of neutralising the coronavirus in patients in an hour, instead of the usual several days. The test can be used to see if potential vaccines work, among other things.
PHOTO: GENSCRIPT BIOTECH CORPORATION
Duke-NUS Medical School has come up with a test kit that takes just an hour, instead of the usual several days, to detect if someone has antibodies which can neutralise the coronavirus that causes Covid-19.
The new test, which The Straits Times first reported on earlier this month, can be used to see if potential vaccines work, to check what proportion of the population has already been infected and for contact tracing, which is critical as Singapore eases up on circuit breaker measures.
Known as cPass, the first-of-its-kind test is available to hospitals here.
When someone is infected with the virus, the body produces hundreds, if not thousands, of different antibodies, which bind with the virus and are known as binding antibodies. However, not all of them can neutralise the virus. This is the role of neutralising antibodies, which bind with proteins on the virus' shell, preventing it from attaching itself to a person's cells.
There are currently Covid-19 tests for such antibodies, but they require the use of a live virus, cells, highly skilled operators, and complex laboratory procedures that require several days to obtain results.
In contrast, the cPass, a blood test, mimics key parts of the testing process chemically and does not require the use of a live virus or cells, said Duke-NUS' Professor Wang Linfa, at a virtual press conference yesterday. It can be carried out in most research or clinical labs, he added.
Prof Wang, who led the team that invented the test, is director of Duke-NUS' emerging infectious diseases programme. The role of antibodies in granting immunity from Covid-19 is currently still being researched.
But Prof Wang said: "We are in the phase of a pandemic where every nation is discussing an exit strategy... in most cases, neutralising antibodies equal protection, or are the best indicator of protection (from the virus)."
He added: "It's not a perfect indicator or biomarker for protection (from Covid-19), but it's as good as you can get right now… We have to be realistic, either you do nothing or do something. And if you want to do something, cPass is the only one that can do it."
Duke-NUS also announced yesterday that it will be co-developing and manufacturing the kit with biotechnology company GenScript Biotech Corporation and the Agency for Science, Technology and Research's Diagnostics Development Hub (DxD Hub).
There are also plans to partner local biotech companies to increase the production of the test kits.
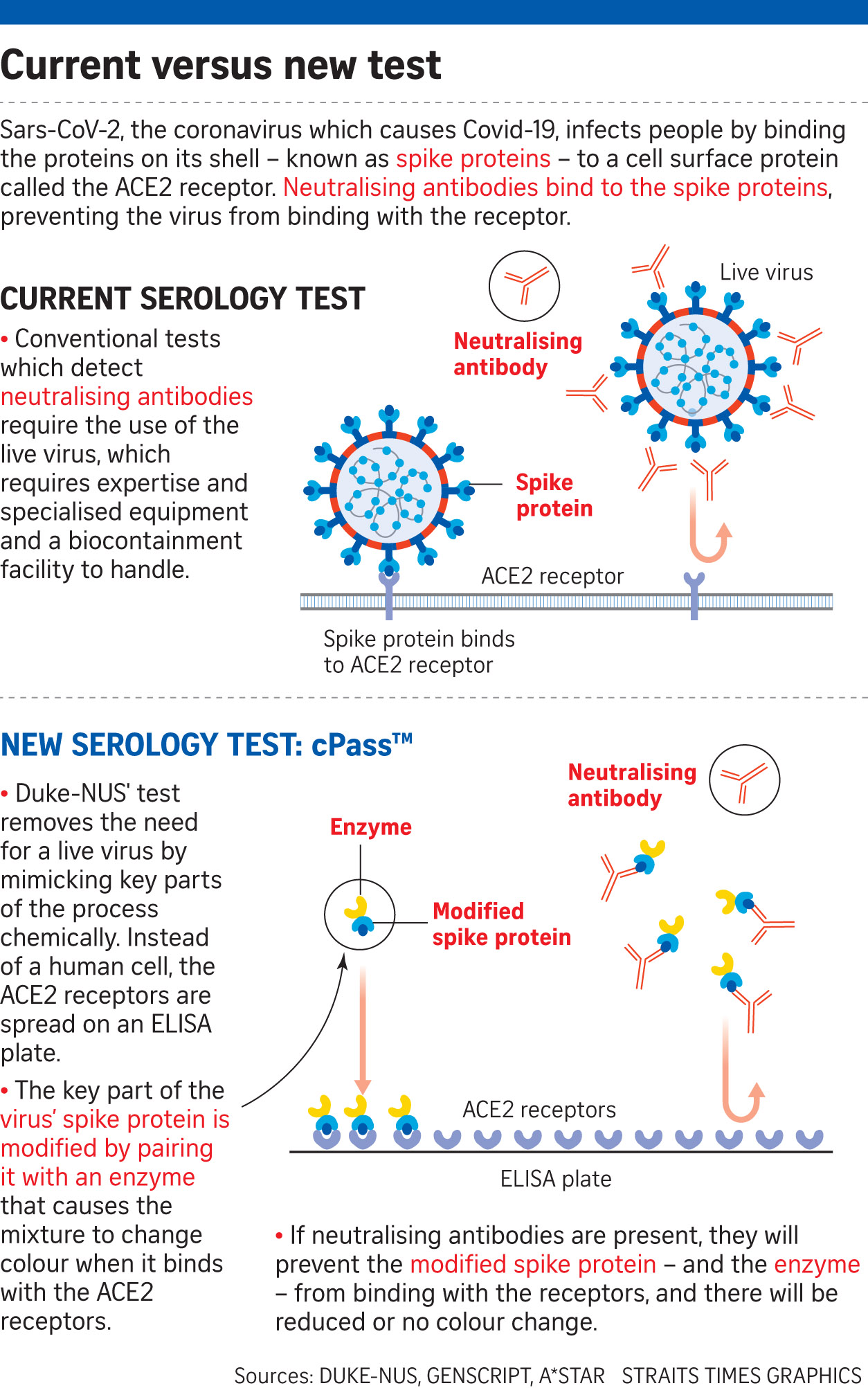
DxD Hub validated the kit with clinical samples of patients, and developed the manufacturing protocol and quality controls to secure its provisional authorisation by the Health Sciences Authority.
GenScript was responsible for proof-of-concept research, product design and development and optimisation, and will now play a central role in the commercialisation process using its global network and manufacturing capacity to launch cPass in Singapore and around the world.
Prof Wang said he came up with the idea for the kit on March 10. Proof of concept data was obtained 10 days later, and by April 24, kits were being shipped to the World Health Organisation's diagnostic labs for assessment.
cPass received provisional authorisation from the Health Sciences Authority on May 8.
"Fighting Covid-19 is like in wartime, where everything has to progress very fast... We could do that due to this three-way partnership," said Prof Wang.
Professor Peter Preiser, associate vice-president (biomedical and life sciences) at Nanyang Technological University, said the test kit would help identify those who were exposed to the virus but did not develop symptoms.
"The ability of this kit to determine the level of neutralising antibodies will provide valuable information on whether a person could be protected against a future infection. This information would be useful to identify the best approach on how to reopen the economy without risking a second wave of infections," he said.
Professor Teo Yik Ying, dean of NUS' Saw Swee Hock School of Public Health, called the test kit a "terrific development".
Knowledge gained from tests conducted by the kit could alter a country's strategies against Covid-19, he said, allowing them to know when to relax their posture.
Prof Teo noted that the test can be deployed in field settings with limited resources.
"This is important for countries where the rural regions typically do not have access to advanced laboratory set-ups," he said.
He added: "Presently there are a few unknowns with regard to whether prior infection immediately confers immunity against re-infection, and even if so, how long this immunity lasts for. This cPass test kit is a crucial resource in allowing the world to learn about these unknowns."


