Singapore-listed Biolidics to distribute Covid-19 rapid test kits in US
Sign up now: Get ST's newsletters delivered to your inbox
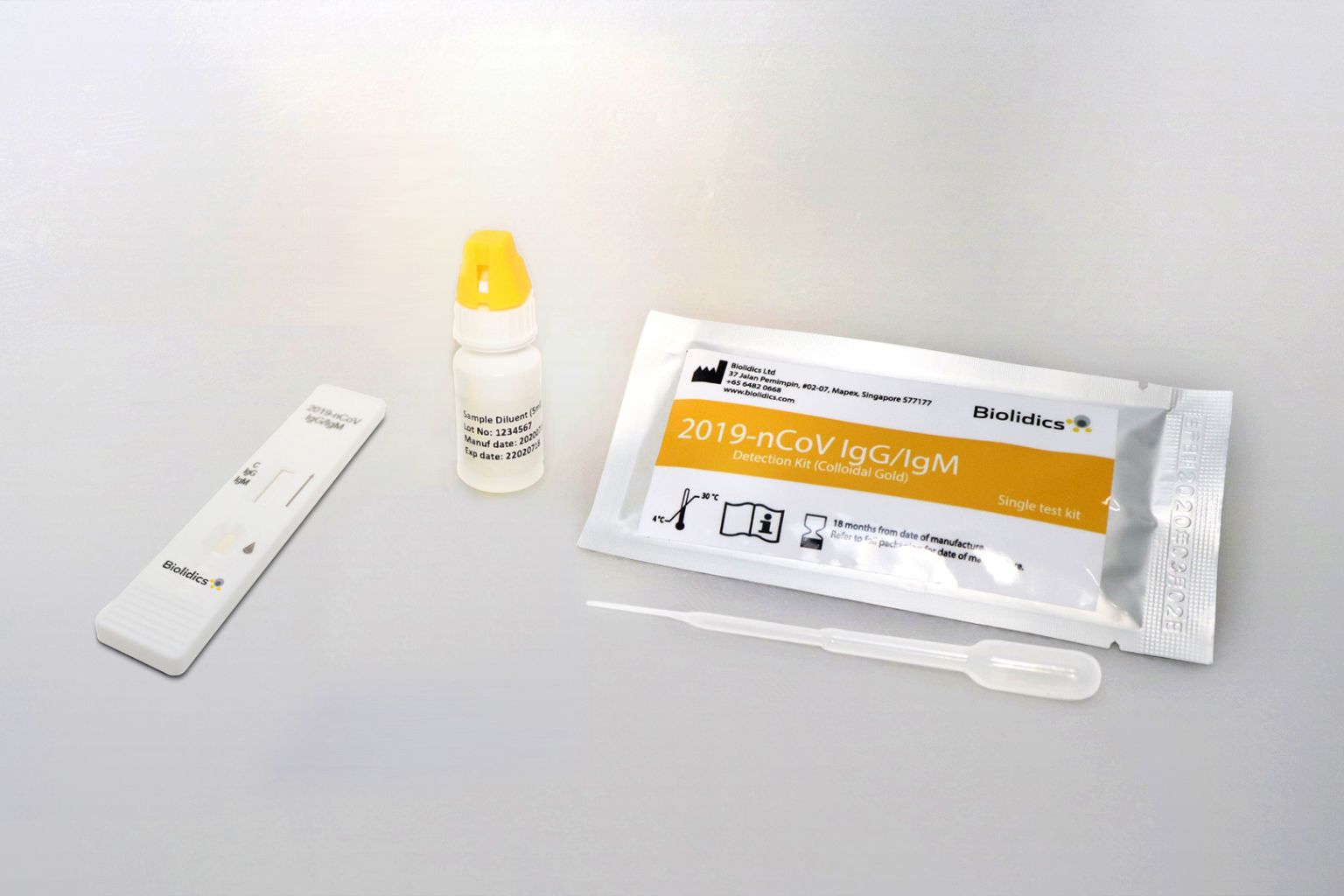
The kits will be allowed for use by clinical laboratories or healthcare workers for point-of-care testing.
PHOTO: BIOLIDICS LIMITED
SINGAPORE (THE BUSINESS TIMES) - Singapore-based cancer diagnostics company Biolidics on Monday (April 13) said it has notified and received acknowledgement from the US Food and Drug Administration (FDA) for the intended distribution of its Covid-19 rapid test kits in the US.
The Catalist-listed firm's kits will be allowed for use by clinical laboratories or healthcare workers for point-of-care testing, but not for home testing.
Biolidics noted that the test kits have not received emergency-use authorisation by the FDA.
The company has to complete the listing of the Covid-19 kits before they can be distributed, marketed and sold. Biolidics is in the process of completing this listing, it said on Monday without specifying a timeline.
For the US, the company will be required to state in its test reports that the tests have not been reviewed by the FDA and that negative results do not rule out the novel coronavirus infection.
The reports must also state that results from antibody testing should not be used as the sole basis to diagnose or exclude Covid-19 infection, and positive results may be due to past or present infection with other coronavirus strains.
Biolidics recently obtained approval to sell its test kits in the European Union and in the Philippines.
In Singapore, the company only disclosed the approval received for the kits after it was certain they could be commercialised, Biolidics said on Thursday in response to the Singapore Exchange's queries. The bourse operator had asked why the firm disclosed this at least a week after the Health Sciences Authority had already announced the information.
Shares of Biolidics closed down 0.5 cent or 1.9 per cent to 26.5 cents on Thursday, before the Good Friday public holiday.


