Indonesia resumes use of temporarily suspended batch of AstraZeneca Covid-19 vaccine
Sign up now: Get insights on Asia's fast-moving developments
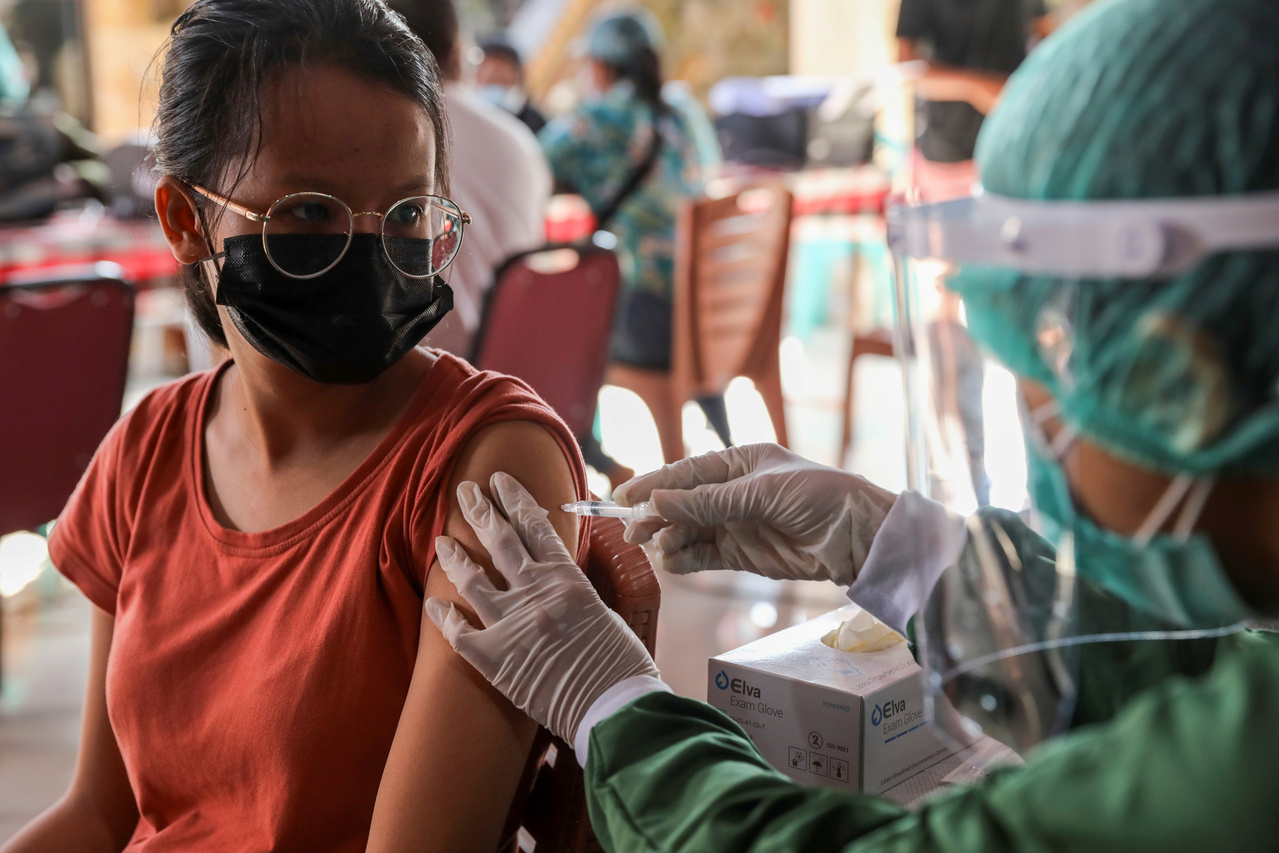
Indonesia had suspended use of one batch of the vaccine to conduct an investigation after a man died.
PHOTO: REUTERS
JAKARTA (REUTERS) - Indonesia has resumed usage of a temporarily suspended batch of AstraZeneca's Covid-19 vaccine, the country's food and drug agency (Bpom) said on Thursday (May 27).
The agency suspended use of one batch of the vaccine to conduct an investigation after a young man died after being inoculated earlier this month.
"According to test results, it can be concluded that there was no relation between the quality of Covid-19 vaccine batch number CTMAV547 with the post immunisation event reported," said Bpom.
The batch consists of 448,480 doses that arrived in the South-east Asian nation in April, part of a delivery of more than 3.85 million doses from the Covax Facility, backed by the World Health Organisation.
Some of the doses have been distributed in Jakarta and the province of North Sulawesi, as well as given to the military, the agency said in a statement.
Indonesia also saw the start of company-funded inoculations for employees in May, with workers in labour-intensive industries such as food and petrochemicals getting their vaccinations first under the roll-out.
Companies taking part in the programme include consumer goods giant Unilever Indonesia, petrochemical producer Chandra Asri and Sinar Mas Agribusiness and Food. Each company will be administering between 2,000 and 4,000 shots for their respective workers.
The private inoculation programme will complement Indonesia's free vaccines for the public. The country started its vaccination drive in mid-January, focusing on high-priority groups such as health workers, the police and the military. This free vaccine programme will officially be expanded to the public by July.


