Philippines to start clinical trials on ivermectin, other drugs for Covid-19
Sign up now: Get insights on Asia's fast-moving developments
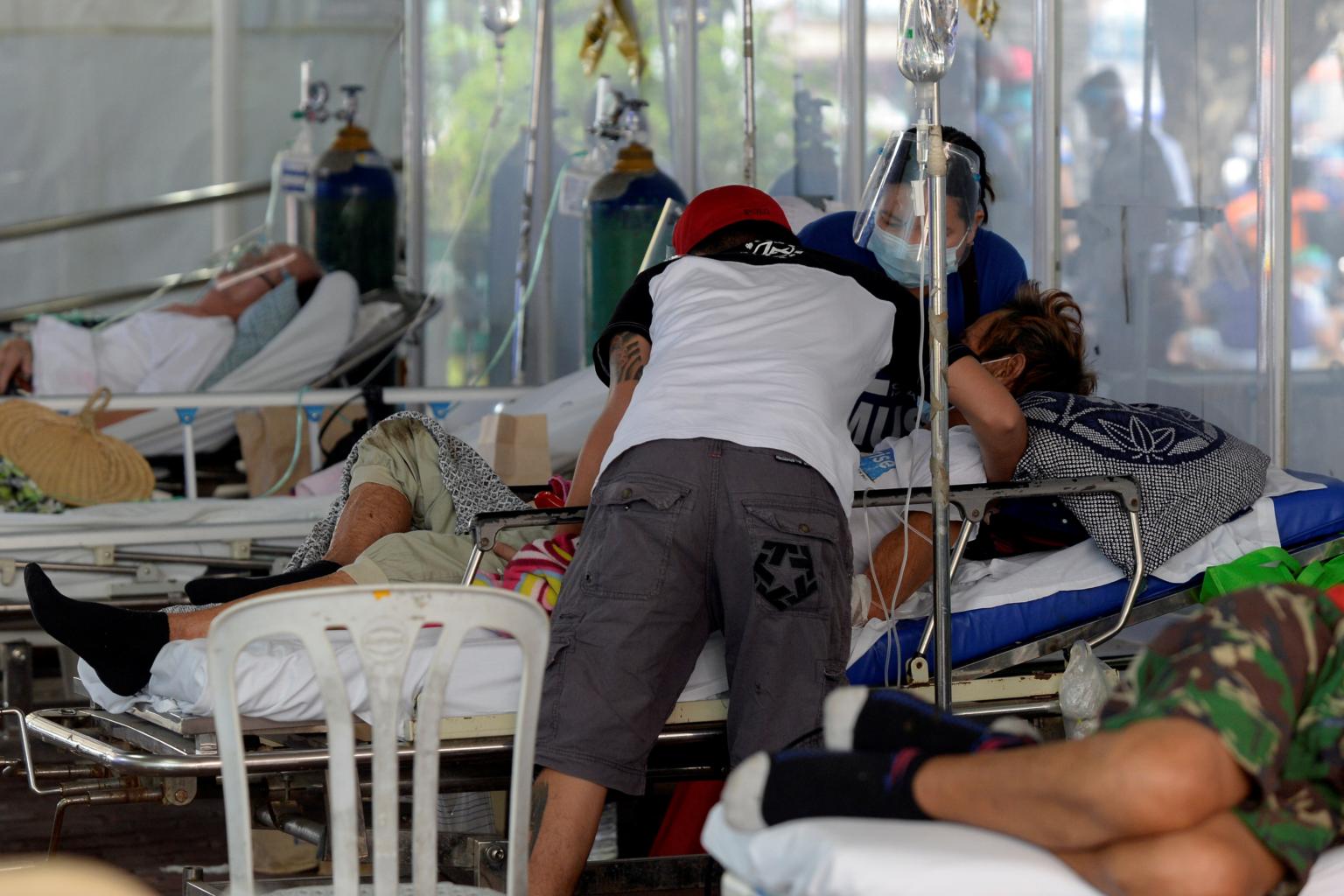
The WHO has warned against using ivermectin in Covid-19 patients except for clinical trials, owing to lack of evidence.
PHOTO: REUTERS
MANILA (REUTERS) - The Philippines will begin clinical trial of several drugs, including the anti-parasite medication ivermectin, in patients with Covid-19 to determine their efficacy in combatting the coronavirus, a senior government official said.
Some politicians in the Philippines have started promoting the use of ivermectin for the coronavirus and given out free doses, although the country's food and drugs regulator has cautioned against its use because of a lack of evidence supporting the use of the drug as a treatment.
The clinical trial for ivermectin, which could last six months, "will give us a more reliable estimate of the effects of ivermectin as an anti-viral agent in mild and moderate (Covid-19) patients", Science and Technology Minister Fortunato Dela Pena said in a presentation late on Monday (April 19).
The South-east Asian nation, which is facing one of the worst coronavirus outbreaks in Asia, is battling a renewed surge in infections, with its vaccination drive reaching only 1.3 million people out of a population of more than 108 million.
Ivermectin tablets have been approved for treating some worm infestations and for veterinary use in animals for parasites.
The World Health Organisation (WHO) last month cautioned against using ivermectin in patients with Covid-19 except for clinical trials, because of a lack of data demonstrating its benefits.
Mr Dela Pena said the government has also approved the clinical trials of a new formulation of methylprednisolone, a steroid, and melatonin, as treatments for Covid-19.
The government will also start trials of an herbal supplement, derived from the native tawa-tawa plant which can fight dengue, he said, adding to ongoing tests using virgin coconut oil for severe Covid-19 patients.
"We are trying several (medications). They may not be vaccines but they could potentially speed up the recovery," Mr Dela Pena said.
The Philippines has recorded more than 945,000 Covid-19 cases and over 16,000 deaths, the second-highest number in South-east Asia, after Indonesia.


