There are more ways to fold proteins than there are atoms in the universe, says Assistant Professor Chester Drum of the National University of Singapore's Yong Loo Lin School of Medicine.
But the assistant professor from the medicine and biochemistry departments has figured out a first - a way to fold a protein using a highly stable shell which wraps around the unfolded substrate, moulding it like hands mould clay, and then protecting it from harm.
Because a protein's shape influences what it can do, this potentially allows Prof Drum and his team to create a tough version of a protein that the body needs, and which could in future help in the treatment of a disease like cancer.
The breakthrough was published in the prestigious journal Nature Communications this month.
The researchers had experimented on a protein in an unusual bacterium - Archaeoglobus fulgidus - that is usually found in hot springs and deep-sea vents living in near-boiling water.
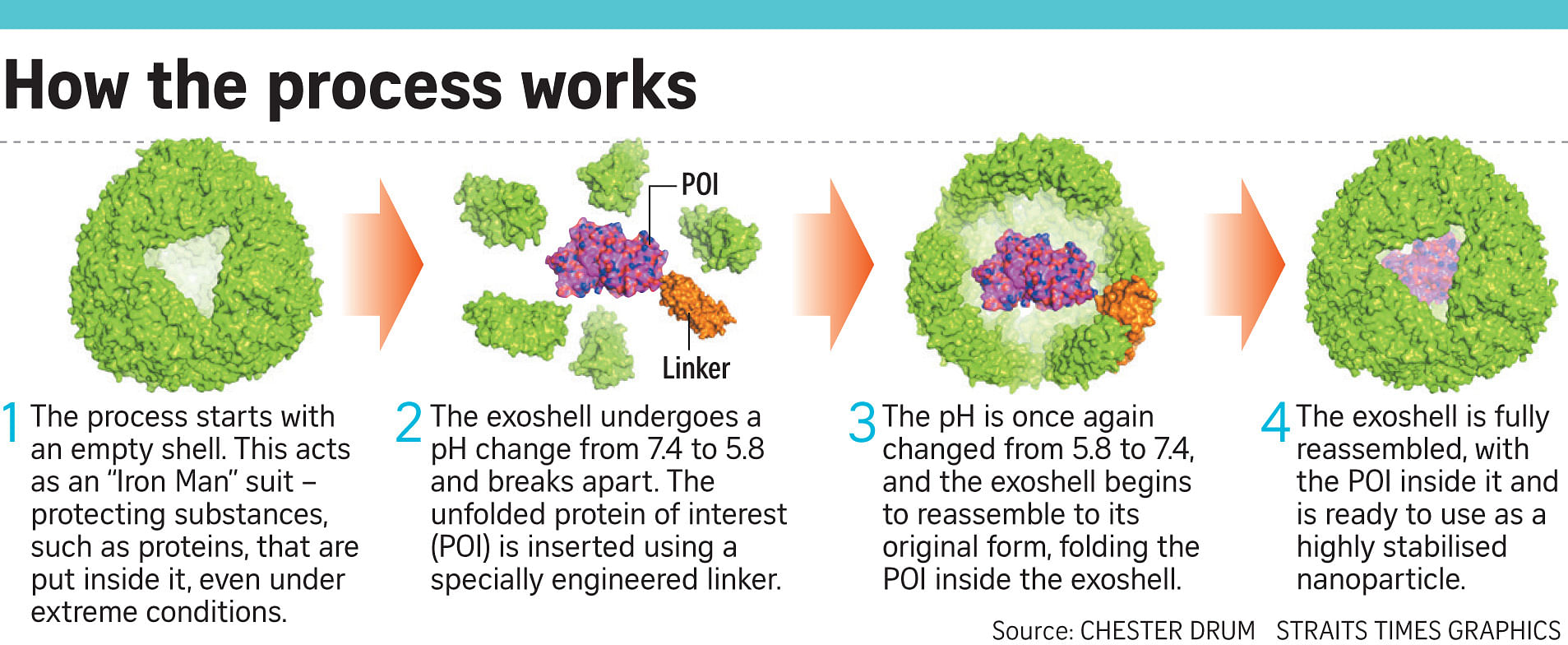
They modified the protein found in the bacterium, called ferritin, to make a nanometre-size exoshell. (A nanometre is one-billionth of a metre.) The protein's natural function is to store iron, but the team re-engineered the natural product to encapsulate a different protein .
This made a Russian-doll-type arrangement where one protein was encased within another.
"We knew that it would be very resilient to our experiments," said Prof Drum, who is also a consultant cardiologist at the National University Hospital. "It's essentially an 'Iron Man' suit, an armour that protects and transports the protein."
In the tests, the scientists first changed the pH level of the shell that they had engineered, breaking it apart. They then inserted the new protein into the shell before changing the pH back to close the shell.
The researchers did this on three different "proteins of interest", showing the technology can be used to fold and protect a variety of proteins over 10,000 times smaller than the width of a human hair.
The exoshells protected the proteins against a wide range of stressors, including extreme heat and harsh chemicals.
"Our highly engineered method means we can specifically guide the folding process, even in a test tube, outside of organs. This makes it more relevant to biological and therapeutic production," said Prof Drum.
The next step, he added, is to work with biopharmaceutical companies to make the exoshell faster and in large quantities. Prof Drum said the exoshell may have the potential to transport substances like antibiotic drugs, or target enzymes and proteins to disease sites in the body, such as an ailing liver or heart.
The team is now using the shell to treat cancer in an experimental animal model and expects to have results within the next year.
Commenting on the effort, Dr Swaine Chen, a senior research scientist at the Agency for Science, Technology and Research's Genome Institute of Singapore, said: "The research shows a very clever application of protein engineering in the realm of synthetic biology, especially with the ability to make these proteins stable."
While the research may not be clinically applicable instantly, it does solve problems with protein production, especially those that are tough to produce in big quantities, he added.