Sinovac included in Singapore's national Covid-19 vaccination programme; 3 doses recommended
Sign up now: Get ST's newsletters delivered to your inbox
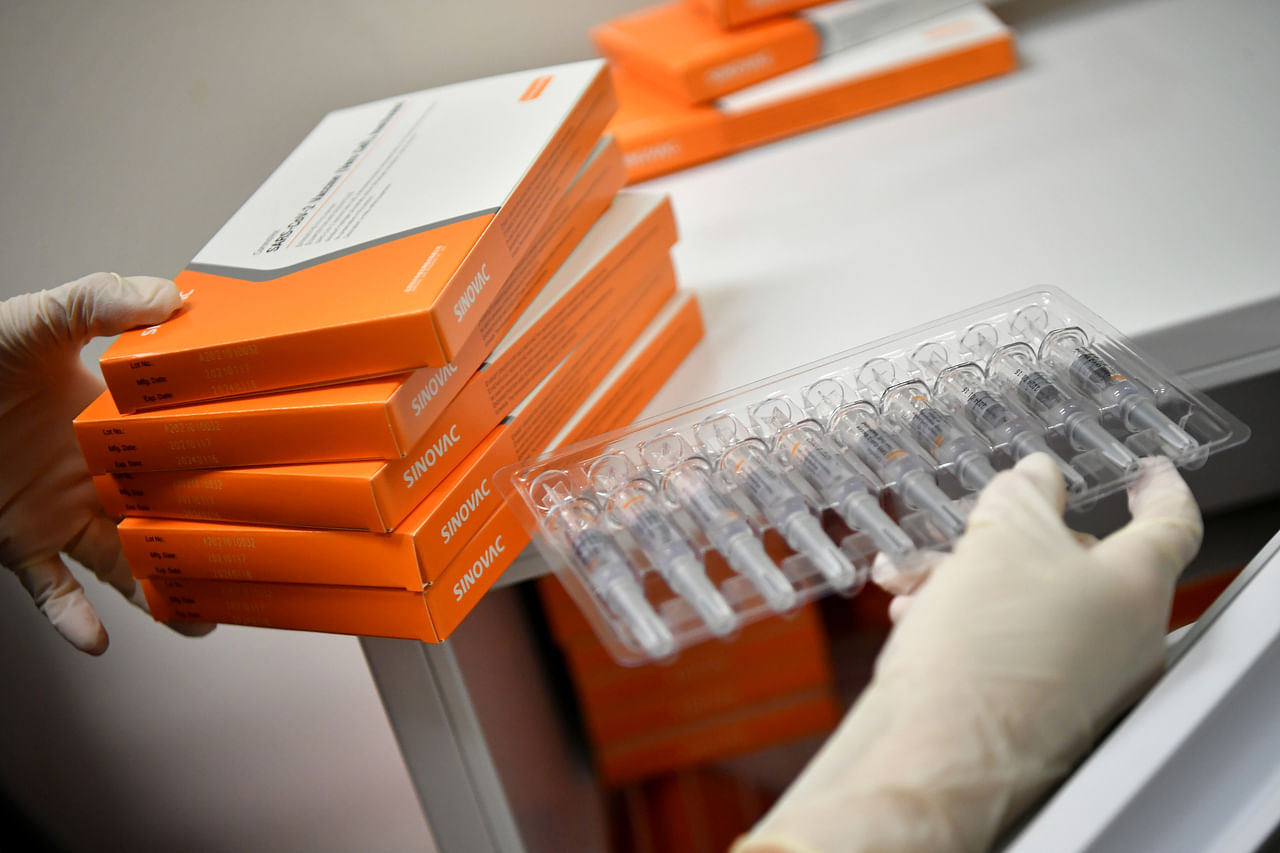
Under the national vaccination programme, the Sinovac vaccine will be administered for free.
ST PHOTO: LIM YAOHUI
Jolene Ang
Follow topic:
SINGAPORE - The Sinovac Covid-19 vaccine will be included in the national vaccination programme to cater to those unable or unwilling to be vaccinated with mRNA vaccines, the multi-ministry task force managing the pandemic said on Saturday (Oct 23).
This follows the Health Sciences Authority's (HSA) interim authorisation of the vaccine under the Pandemic Special Access Route (PSAR).
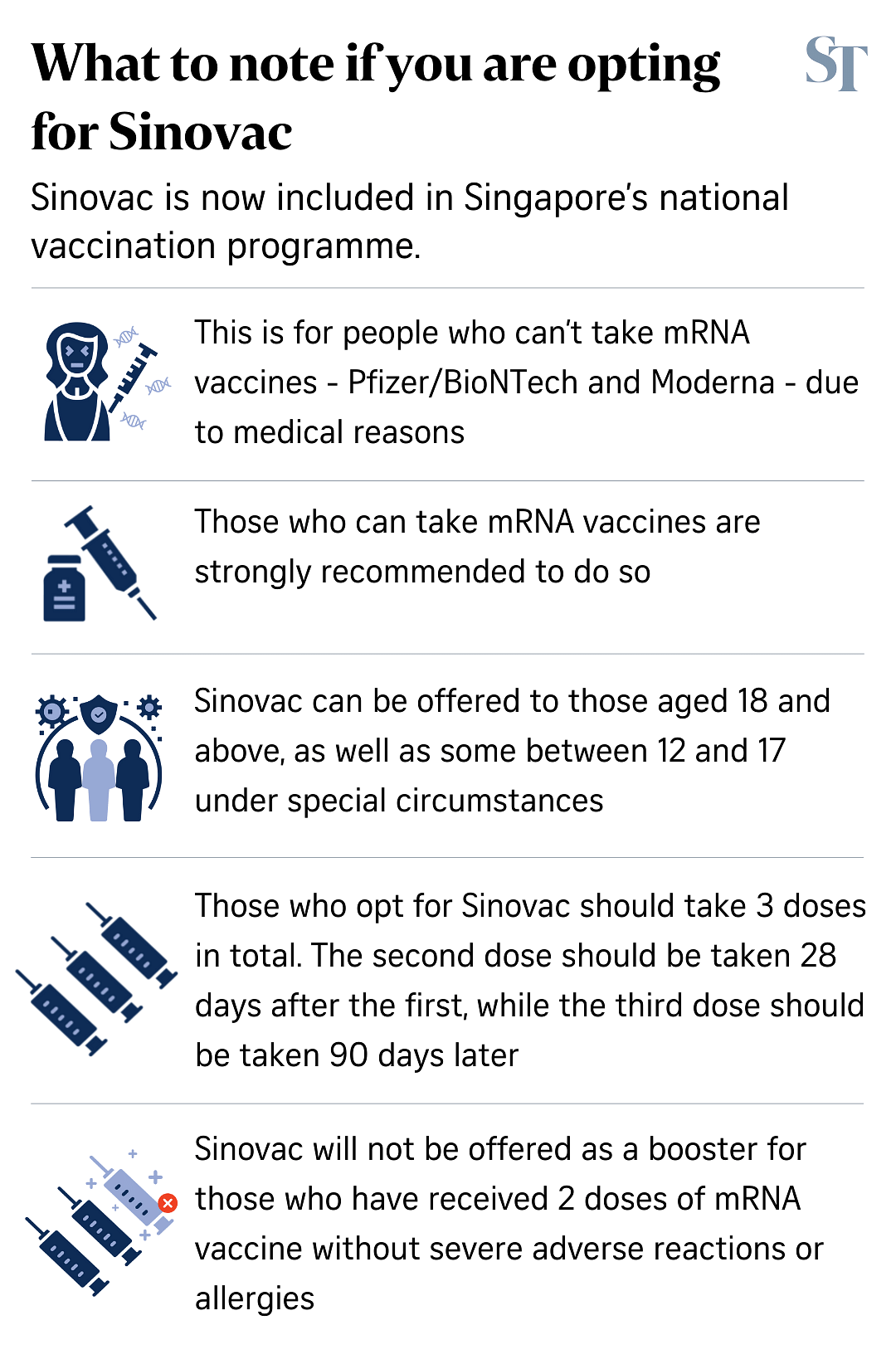
Three doses of the Sinovac vaccine will be required for a person to be considered fully vaccinated, said the Ministry of Health (MOH).
The second dose should be taken 28 days after the first dose, while the third dose should be taken 90 days after the second dose, the HSA said.
All individuals who have taken two doses of the Sinovac vaccine will be regarded as fully vaccinated for four months after the second dose, or Dec 31 this year, whichever is later.
Individuals will have to get their third doses by then in order to maintain their vaccination status.
Under the national vaccination programme, the Sinovac vaccine will be administered for free, and will come under the Vaccine Injury Financial Assistance programme.
However, individuals who took the Sinovac vaccine before PSAR approval was given and paid for it themselves will not be reimbursed, said Health Minister Ong Ye Kung, who co-chairs the taskforce.
MOH will assign the vaccination centre at Raffles City Convention Centre to administer the Sinovac vaccine, Mr Ong added.
This is on top of 11 Public Health Preparedness Clinics as well as select private clinics.
MOH provided the following guidelines on Saturday:
- People who have received only one or two doses of the Sinovac vaccine to date are strongly encouraged to complete a three-dose primary series regime with the mRNA vaccines as it offers more optimal protection.
- People who are unable to take any dose of the mRNA vaccines due to medical reasons should receive three doses of Sinovac in total to complete their primary series regime.
- Those who started vaccination with mRNA vaccines but developed allergies or severe adverse reactions after the first dose should complete a three-dose primary series regime with two more doses of Sinovac.
- If the allergic reaction happens after the second dose of mRNA vaccine, then the person should take one more dose of Sinovac as their booster vaccination when they are eligible for a booster.
- Sinovac will be not offered as a booster for people who have already completed two doses of mRNA vaccine and did not develop allergies or severe adverse reactions. For this group, a third dose of mRNA vaccine will be offered to them as the booster dose instead under the National Vaccination Programme.
Mr Ong said that of the eligible population, 94 per cent have either taken or have booked their vaccinations.
But among the 6 per cent of the eligible population who remain unvaccinated, 70,000 are seniors above 70 years or 60 years old.
"They are at a high risk of falling very sick if infected with Covid-19," Mr Ong said.
"Some of these unvaccinated individuals are unable to take the mRNA vaccines due to medical reasons. Others may just simply prefer a non-mRNA vaccine.
"So we will make the Sinovac vaccine more accessible to them, to encourage them to get vaccinated."
The ministry also said that the Sinovac vaccine has not been approved for general use for individuals aged 12 to 17 years.
This is not in the application from Sinovac and is also consistent with the World Health Organisation's approved use under the Emergency Use Listing.
However, for those in this age band who are medically ineligible to complete a full two-dose regime of the Pfizer-BioNTech/Comirnaty mRNA vaccines, MOH will offer a three-dose primary series using Sinovac under a dedicated public health programme.
They will be closely monitored by trained medical personnel under this public health programme, given that the use of Sinovac in persons under the age of 18 years was not included in HSA's PSAR interim authorisation, said MOH.
"We will get in touch with eligible persons in this age group on how they can receive the Sinovac vaccine safely."
Addressing a question at Saturday's press conference on why the Sinopharm vaccine is not being included in the national vaccination programme, Mr Ong said "there has been no application from Sinopharm".
"We had an application with Sinovac... It's been given interim approval. The number of doses being administered in Singapore is not big enough for us to do a comprehensive study, not for Sinovac, and for Sinopharm, it is even lower."
But studies in Brazil and Chile have yielded some good data that helps in the comparisons of vaccine effectiveness against infection, severe illnesses and death, he noted.

