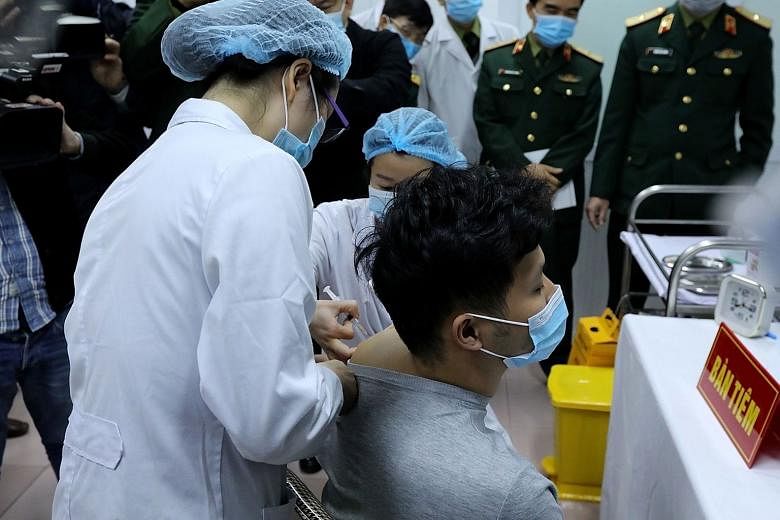
HANOI • Vietnam yesterday began human trials of its Covid-19 vaccine, with three volunteers receiving their first doses of Nanocovax at the Military Medical University in the capital Hanoi.
The two men and a woman, whose identities have been kept private, will be monitored for 72 hours at the academy before being allowed to return to their homes. They will be monitored for 56 days after the vaccination.
The three were chosen from about 300 who volunteered for the human trials of the vaccine, developed by Nanogen Pharmaceutical Biotechnology JSC which is based in Ho Chi Minh City.
Lieutenant-General Do Quyet, director of the medical university, said: "It's time Vietnam can proclaim to the world that we can do this, just like the evident successes we have had in preventing and controlling Covid-19 in the country."
Vietnam has recorded very low coronavirus infection rates, with only 1,405 cases and 35 deaths reported so far.
Its government is committed to producing a homegrown coronavirus vaccine to avoid reliance on other vaccines whose supply might not be sufficient to meet worldwide demand.
"The stock of approved vaccines can only reach one-fifth of the global population and most of it will go to developed countries," said Dr Quyet at a press briefing after the vaccine was administered to the three volunteers.
The first phase trial for Nanocovax is expected to last four months. This will be followed by another four-month trial for the second phase which will involve 400 to 600 test subjects, and six months for the third and final phase which might involve as many as 10,000 people in Vietnam, Bangladesh, India and Indonesia, according to the Military Medical University.
Nanogen's director of research and development Do Minh Tri said the company has worked out a special health insurance plan to cover the health risks taken by the vaccine volunteers, with the payment set at 100 million dong (S$5,700) per incident.
The clinical trial results will not be available until the end of next year.
Nanocovax is the first Vietnam-made Covid-19 vaccine to enter human trials, with another two from other manufacturers to follow in February and March. Nanogen said it could produce five million doses per month.
Nanocovax can be stored in normal cold storage at 2-8 deg C.
With state subsidies, the price of each injection is set at 120,000 dong and each person will need two shots, with an interval of 28 days between the shots.
VIETNAM NEWS/ASIA NEWS NETWORK