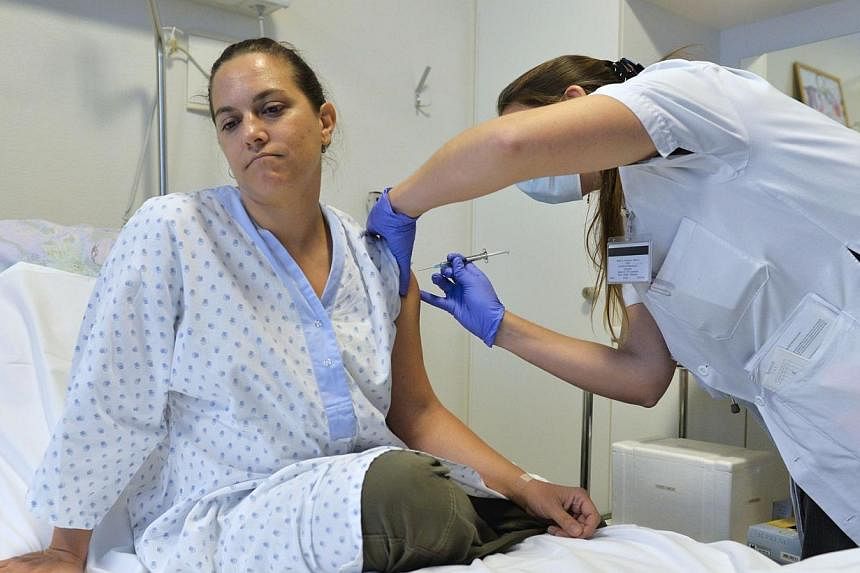
GENEVA (Reuters) - A clinical trial of the Ebola vaccine by Merck and NewLink has been interrupted in all 59 volunteers in Geneva a week early "as a measure of precaution" after four patients complained of joint pains in hands and feet, the University of Geneva Hospital said on Thursday.
"They are all fine and being monitored regularly by the medical team leading the study," it said in a statement.
The human safety trials are due to resume on Jan. 5 in up to 15 volunteers after checks to ensure that the joint pain symptoms were "benign and temporary", the hospital said.